Inhibition of NF-kappaB signaling reduces virus load and gammaherpesvirus-induced pulmonary fibrosis
- PMID: 20566741
- PMCID: PMC2913377
- DOI: 10.2353/ajpath.2010.091122
Inhibition of NF-kappaB signaling reduces virus load and gammaherpesvirus-induced pulmonary fibrosis
Abstract
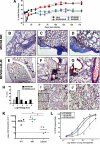
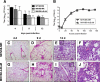
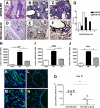
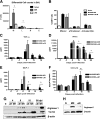
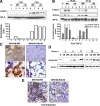
Idiopathic pulmonary fibrosis (IPF) is a chronic progressive lung disorder of unknown etiology. Several studies have demonstrated an association between pulmonary infection with a herpesvirus and IPF. Based on those observations, we have developed a mouse model in which interferon (IFN)gammaR(-/-) mice infected intranasally with murine gammaherpesvirus 68 (MHV68) develop lung fibrosis. We hypothesize that viral load was a critical factor for the development of fibrosis. Because nuclear factor (NF)-kappaB signaling is required to efficiently establish gammaherpesvirus, latency we infected IFNgammaR(-/-) mice with a MHV68 virus that expresses a mutant dominant inhibitor of the NF-kappaB signaling pathway, called IkappaBalphaM. Striking differences were observed at the onset of the chronic infection, which correlated with a decreased virus load in mice infected with MHV68-IkappaBalphaM compared with mice infected with control MHV68 (MHV68-MR). IFNgammaR(-/-) mice infected with MHV68-IkappaBalphaM lacked vasculitis and fibrosis 15 to 120 days post infection. Inhibition of NF-kappaB in MHV68-infected cells of the lungs diminished the expression of the fibrocyte recruiting chemokines monocyte chemoattractant protein 1 (MCP-1) and CXCL12, ameliorated macrophage expression of markers of alternative activation, and failed to increase expression of the integrin alphavbeta6, which is implicated in the activation of the profibrotic factor TGF-beta. Thus, the inhibition of NF-kappaB signaling in the infected lung cells of IFNgammaR(-/-) mice reduces virus persistence and ameliorates profibrotic events. Host determinants of latency might therefore represent new therapeutic targets for gammaherpesvirus-associated pulmonary fibrosis.
Figures

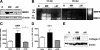
Similar articles
-
CD8+ T Cell Response to Gammaherpesvirus Infection Mediates Inflammation and Fibrosis in Interferon Gamma Receptor-Deficient Mice.PLoS One. 2015 Aug 28;10(8):e0135719. doi: 10.1371/journal.pone.0135719. eCollection 2015. PLoS One. 2015. PMID: 26317335 Free PMC article.
-
Control of virus reactivation arrests pulmonary herpesvirus-induced fibrosis in IFN-gamma receptor-deficient mice.Am J Respir Crit Care Med. 2007 Jun 1;175(11):1139-50. doi: 10.1164/rccm.200610-1426OC. Epub 2007 Mar 15. Am J Respir Crit Care Med. 2007. PMID: 17363768 Free PMC article.
-
IKKα-Mediated Noncanonical NF-κB Signaling Is Required To Support Murine Gammaherpesvirus 68 Latency In Vivo.J Virol. 2022 May 25;96(10):e0002722. doi: 10.1128/jvi.00027-22. Epub 2022 Apr 28. J Virol. 2022. PMID: 35481781 Free PMC article.
-
Gammaherpesviruses and pulmonary fibrosis: evidence from humans, horses, and rodents.Vet Pathol. 2014 Mar;51(2):372-84. doi: 10.1177/0300985814521838. Vet Pathol. 2014. PMID: 24569614 Review.
-
Murine Gammaherpesvirus 68: A Small Animal Model for Gammaherpesvirus-Associated Diseases.Adv Exp Med Biol. 2017;1018:225-236. doi: 10.1007/978-981-10-5765-6_14. Adv Exp Med Biol. 2017. PMID: 29052141 Review.
Cited by
-
Role of endoplasmic reticulum stress in age-related susceptibility to lung fibrosis.Am J Respir Cell Mol Biol. 2012 Jun;46(6):748-56. doi: 10.1165/rcmb.2011-0224OC. Epub 2012 Jan 6. Am J Respir Cell Mol Biol. 2012. PMID: 22227563 Free PMC article.
-
CD8+ T Cell Response to Gammaherpesvirus Infection Mediates Inflammation and Fibrosis in Interferon Gamma Receptor-Deficient Mice.PLoS One. 2015 Aug 28;10(8):e0135719. doi: 10.1371/journal.pone.0135719. eCollection 2015. PLoS One. 2015. PMID: 26317335 Free PMC article.
-
Nuclear factor-κB: a key regulator in health and disease of lungs.Arch Immunol Ther Exp (Warsz). 2011 Oct;59(5):335-51. doi: 10.1007/s00005-011-0136-z. Epub 2011 Jul 26. Arch Immunol Ther Exp (Warsz). 2011. PMID: 21786215 Free PMC article. Review.
-
Conquering the Host: Determinants of Pathogenesis Learned from Murine Gammaherpesvirus 68.Annu Rev Virol. 2021 Sep 29;8(1):349-371. doi: 10.1146/annurev-virology-011921-082615. Annu Rev Virol. 2021. PMID: 34586873 Free PMC article.
-
Tiled microarray identification of novel viral transcript structures and distinct transcriptional profiles during two modes of productive murine gammaherpesvirus 68 infection.J Virol. 2012 Apr;86(8):4340-57. doi: 10.1128/JVI.05892-11. Epub 2012 Feb 8. J Virol. 2012. PMID: 22318145 Free PMC article.
References
-
- American Thoracic Society Idiopathic pulmonary fibrosis: diagnosis and treatment. International consensus statement. American Thoracic Society (ATS), and the European Respiratory Society (ERS). Am J Respir Crit Care Med. 2000;161:646–664. - PubMed
-
- Stewart JP, Egan JJ, Ross AJ, Kelly BG, Lok SS, Hasleton PS, Woodcock AA. The detection of Epstein-Barr virus DNA in lung tissue from patients with idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 1999;159:1336–1341. - PubMed
-
- Kelly BG, Lok SS, Hasleton PS, Egan JJ, Stewart JP. A rearranged form of Epstein-Barr virus DNA is associated with idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2002;166:510–513. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

