Localization-dependent activity of the Kv2.1 delayed-rectifier K+ channel
- PMID: 20566856
- PMCID: PMC2901471
- DOI: 10.1073/pnas.1003028107
Localization-dependent activity of the Kv2.1 delayed-rectifier K+ channel
Abstract
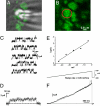
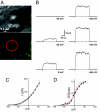
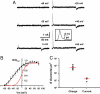
The Kv2.1 K(+) channel is highly expressed throughout the brain, where it regulates excitability during periods of high-frequency stimulation. Kv2.1 is unique among Kv channels in that it targets to large surface clusters on the neuronal soma and proximal dendrites. These clusters also form in transfected HEK cells. Following excessive excitatory stimulation, Kv2.1 declusters with an accompanying 20- to 30-mV hyperpolarizing shift in the activation threshold. Although most Kv2.1 channels are clustered, there is a pool of Kv2.1 resident outside of these domains. Using the cell-attached patch clamp technique, we investigated the hypothesis that Kv2.1 activity varies as a function of cell surface location. We found that clustered Kv2.1 channels do not efficiently conduct K(+), whereas the nonclustered channels are responsible for the high threshold delayed rectifier K(+) current typical of Kv2.1. Comparison of gating and ionic currents indicates only 2% of the surface channels conduct, suggesting that the clustered channels still respond to membrane potential changes. Declustering induced via either actin depolymerization or alkaline phosphatase treatment did not increase whole-cell currents. Dephosphorylation resulted in a 25-mV hyperpolarizing shift, whereas actin depolymerization did not alter the activation midpoint. Taken together, these data demonstrate that clusters do not contain high threshold Kv2.1 channels whose voltage sensitivity shifts upon declustering; nor are they a reservoir of nonconducting channels that are activated upon release. On the basis of these findings, we propose unique roles for the clustered Kv2.1 that are independent of K(+) conductance.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Misonou H, Trimmer JS. Determinants of voltage-gated potassium channel surface expression and localization in mammalian neurons. Crit Rev Biochem Mol Biol. 2004;39:125–145. - PubMed
-
- Tamarina NA, Kuznetsov A, Fridlyand LE, Philipson LH. Delayed-rectifier (KV2.1) regulation of pancreatic beta-cell calcium responses to glucose: Inhibitor specificity and modeling. Am J Physiol Endocrinol Metab. 2005;289:E578–E585. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

