Single-cell analysis of insulin-regulated fatty acid uptake in adipocytes
- PMID: 20570821
- PMCID: PMC2944284
- DOI: 10.1152/ajpendo.00330.2010
Single-cell analysis of insulin-regulated fatty acid uptake in adipocytes
Abstract
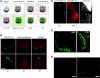
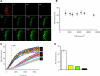
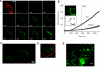
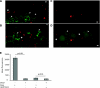
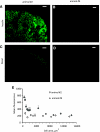
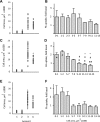
Increased body fat correlates with the enlargement of average fat cell size and reduced adipose tissue insulin sensitivity. It is currently unclear whether adipocytes, as they accumulate more triglycerides and grow in size, gradually become less insulin sensitive or whether obesity-related factors independently cause both the enlargement of adipocyte size and reduced adipose tissue insulin sensitivity. In the first instance, large and small adipocytes in the same tissue would exhibit differences in insulin sensitivity, whereas, in the second instance, adipocyte size per se would not necessarily correlate with insulin response. To analyze the effect of adipocyte size on insulin sensitivity, we employed a new single-cell imaging assay that resolves fatty acid uptake and insulin response in single adipocytes in subcutaneous adipose tissue explants. Here, we report that subcutaneous adipocytes are heterogeneous in size and intrinsic insulin sensitivity. Whereas smaller adipocytes respond to insulin by increasing lipid uptake, adipocytes with cell diameters larger than 80-100 microm are insulin resistant. We propose that, when cell size approaches a critical boundary, adipocytes lose insulin-dependent fatty acid transport. This negative feedback mechanism may protect adipocytes from lipid overload and restrict further expansion of adipose tissue, which leads to obesity and metabolic complications.
Figures
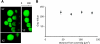
References
-
- Abel ED, Peroni O, Kim JK, Kim YB, Boss O, Hadro E, Minnemann T, Shulman GI, Kahn BB. Adipose-selective targeting of the GLUT4 gene impairs insulin action in muscle and liver. Nature 409: 729–733, 2001 - PubMed
-
- Agarwal AK, Garg A. Genetic disorders of adipose tissue development, differentiation, and death. Annu Rev Genomics Hum Genet 7: 175–199, 2006 - PubMed
-
- Ahima RS. Adipose tissue as an endocrine organ. Obesity (Silver Spring) 14, Suppl 5: 242S–249S, 2006 - PubMed
-
- Bergman RN, Ader M. Free fatty acids and pathogenesis of type 2 diabetes mellitus. Trends Endocrinol Metab 11: 351–356, 2000 - PubMed
-
- Berk PD, Zhou SL, Kiang CL, Stump D, Bradbury M, Isola LM. Uptake of long chain free fatty acids is selectively up-regulated in adipocytes of Zucker rats with genetic obesity and non-insulin-dependent diabetes mellitus. J Biol Chem 272: 8830–8835, 1997 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

