Alternatively spliced RAGEv1 inhibits tumorigenesis through suppression of JNK signaling
- PMID: 20570900
- PMCID: PMC2919303
- DOI: 10.1158/0008-5472.CAN-10-0595
Alternatively spliced RAGEv1 inhibits tumorigenesis through suppression of JNK signaling
Abstract
Receptor for advanced glycation end products (RAGE) and its ligands are overexpressed in multiple cancers. RAGE has been implicated in tumorigenesis and metastasis, but little is known of the mechanisms involved. In this study, we define a specific functional role for an alternate splice variant termed RAGE splice variant 1 (RAGEv1), which encodes a soluble endogenous form of the receptor that inhibits tumorigenesis. RAGEv1 was downregulated in lung, prostate, and brain tumors relative to control matched tissues. Overexpressing RAGEv1 in tumor cells altered RAGE ligand stimulation of several novel classes of genes that are critical in tumorigenesis and metastasis. Additionally, RAGEv1 inhibited tumor formation, cell invasion, and angiogenesis induced by RAGE ligand signaling. Analysis of signal transduction pathways underlying these effects revealed marked suppression of c-jun-NH(2)-kinase (JNK) pathway signaling, and JNK inhibition suppressed signaling through the RAGE pathway. Tumors expressing RAGEv1 were significantly smaller than wild-type tumors and displayed prominently reduced activation of JNK. Our results identify RAGEv1 as a novel suppressor, the study of which may offer new cancer therapeutic directions.
Copyright 2010 AACR.
Figures
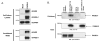

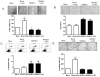
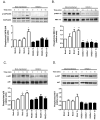
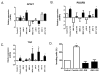

References
-
- Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000 Jan 7;100(1):57–70. - PubMed
-
- Taguchi A, Blood DC, del Toro G, Canet A, Lee DC, Qu W, et al. Blockage of RAGE-amphoterin signalling suppresses tumour growth and metastases. Nature. 2000;405:354–60. - PubMed
-
- Logsdon CD, Fuentes MK, Huang EH, Arumugam T. RAGE and RAGE ligands in cancer. Curr Mol Med. 2007 Dec;7(8):777–89. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

