Robust in vivo transduction of nervous system and neural stem cells by early gestational intra amniotic gene transfer using lentiviral vector
- PMID: 20571539
- PMCID: PMC2956924
- DOI: 10.1038/mt.2010.125
Robust in vivo transduction of nervous system and neural stem cells by early gestational intra amniotic gene transfer using lentiviral vector
Abstract
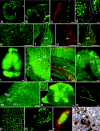
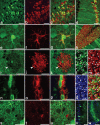
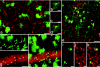
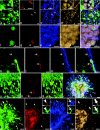
Presently, in vivo methods to efficiently and broadly transduce all major cell types throughout both the central (CNS) and peripheral adult nervous system (PNS) are lacking. In this study, we hypothesized that during early fetal development neural cell populations, including neural stem cells (NSCs), may be accessible for gene transfer via the open neural groove. To test this hypothesis, we injected lentiviral vectors encoding a green fluorescent protein (GFP) marker gene into the murine amniotic cavity at embryonic day 8. This method (i) efficiently and stably transduced the entire nervous system for at least 80% of the lifespan of the mice, (ii) transduced all major neural cell types, and (iii) transduced adult NSCs of the subventricular zone (SVZ) and subgranular zones (SGZs). This simple approach has broad applications for the study of gene function in nervous system development and adult NSCs and may have future clinical applications for treatment of genetic disorders of the nervous system.
Figures
Similar articles
-
Efficient in vivo targeting of epidermal stem cells by early gestational intraamniotic injection of lentiviral vector driven by the keratin 5 promoter.Mol Ther. 2008 Jan;16(1):131-7. doi: 10.1038/sj.mt.6300332. Epub 2007 Oct 9. Mol Ther. 2008. PMID: 17923841 Free PMC article.
-
Gene transfer into the central nervous system in vivo using a recombinanat lentivirus vector.J Neurosci Res. 2002 Feb 1;67(3):363-71. doi: 10.1002/jnr.10137. J Neurosci Res. 2002. PMID: 11813241
-
Efficient transduction of neurons using Ross River glycoprotein-pseudotyped lentiviral vectors.Gene Ther. 2006 Jun;13(12):966-73. doi: 10.1038/sj.gt.3302701. Epub 2006 Mar 2. Gene Ther. 2006. PMID: 16511527
-
In vivo transduction of murine cerebellar Purkinje cells by HIV-derived lentiviral vectors.Brain Res. 2006 Apr 12;1082(1):11-22. doi: 10.1016/j.brainres.2006.01.104. Epub 2006 Mar 6. Brain Res. 2006. PMID: 16516872
-
Gene transfer to ocular stem cells by early gestational intraamniotic injection of lentiviral vector.Mol Ther. 2007 Mar;15(3):579-87. doi: 10.1038/sj.mt.6300092. Epub 2007 Jan 23. Mol Ther. 2007. PMID: 17245352
Cited by
-
Surface conjugation of antibodies improves nanoparticle uptake in bronchial epithelial cells.PLoS One. 2022 Apr 6;17(4):e0266218. doi: 10.1371/journal.pone.0266218. eCollection 2022. PLoS One. 2022. PMID: 35385514 Free PMC article.
-
Amniotic fluid stabilized lipid nanoparticles for in utero intra-amniotic mRNA delivery.J Control Release. 2022 Jan;341:616-633. doi: 10.1016/j.jconrel.2021.10.031. Epub 2021 Nov 3. J Control Release. 2022. PMID: 34742747 Free PMC article.
-
Developmental stage determines efficiency of gene transfer to muscle satellite cells by in utero delivery of adeno-associated virus vector serotype 2/9.Mol Ther Methods Clin Dev. 2014 Sep 10;1:14040. doi: 10.1038/mtm.2014.40. eCollection 2014. Mol Ther Methods Clin Dev. 2014. PMID: 26015979 Free PMC article.
-
G-protein-coupled receptors in adult neurogenesis.Pharmacol Rev. 2012 Jul;64(3):645-75. doi: 10.1124/pr.111.004762. Epub 2012 May 18. Pharmacol Rev. 2012. PMID: 22611178 Free PMC article. Review.
-
Genetic therapy for the nervous system.Hum Mol Genet. 2011 Apr 15;20(R1):R28-41. doi: 10.1093/hmg/ddr110. Epub 2011 Mar 23. Hum Mol Genet. 2011. PMID: 21429918 Free PMC article. Review.
References
-
- Abrous DN, Koehl M., and, Le Moal M. Adult neurogenesis: from precursors to network and physiology. Physiol Rev. 2005;85:523–569. - PubMed
-
- Alvarez-Buylla A, García-Verdugo JM., and, Tramontin AD. A unified hypothesis on the lineage of neural stem cells. Nat Rev Neurosci. 2001;2:287–293. - PubMed
-
- Gage FH. Mammalian neural stem cells. Science. 2000;287:1433–1438. - PubMed
-
- Gaiano N, Kohtz JD, Turnbull DH., and, Fishell G. A method for rapid gain-of-function studies in the mouse embryonic nervous system. Nat Neurosci. 1999;2:812–819. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

