Triggering N(2) uptake via redox-induced expulsion of coordinated NH(3) and N(2) silylation at trigonal bipyramidal iron
- PMID: 20571574
- PMCID: PMC2892393
- DOI: 10.1038/nchem.660
Triggering N(2) uptake via redox-induced expulsion of coordinated NH(3) and N(2) silylation at trigonal bipyramidal iron
Abstract
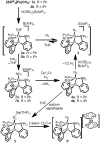
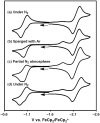
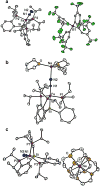
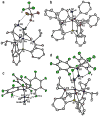
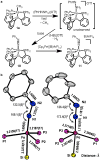
The biological reduction of N(2) to give NH(3) may occur by one of two predominant pathways in which nitrogenous N(x)H(y) intermediates, including hydrazine (N(2)H(4)), diazene (N(2)H(2)), nitride (N(3-)) and imide (NH(2-)), may be involved. To test the validity of hypotheses on iron's direct role in the stepwise reduction of N(2), model systems for iron are needed. Such systems can test the chemical compatibility of iron with various proposed N(x)H(y) intermediates and the reactivity patterns of such species. Here we describe a trigonal bipyramidal Si(o-C(6)H(4)PR(2))(3)Fe-L scaffold (R = Ph or i-Pr) in which the apical site is occupied by nitrogenous ligands such as N(2), N(2)H(4), NH(3) and N(2)R. The system accommodates terminally bound N(2) in the three formal oxidation states (iron(0), +1 and +2). N(2) uptake is demonstrated by the displacement of its reduction partners NH(3) and N(2)H(4), and N(2) functionalizaton is illustrated by electrophilic silylation.
Figures


Similar articles
-
Intermediates in the reduction of N(2) to NH(3): synthesis of iron eta(2) hydrazido(1-) and diazene complexes.Dalton Trans. 2009 Jun 21;(23):4420-2. doi: 10.1039/b902524c. Epub 2009 Apr 14. Dalton Trans. 2009. PMID: 19488434
-
A five-coordinate phosphino/acetate iron(II) scaffold that binds N2, N2H2, N2H4, and NH3 in the sixth site.Inorg Chem. 2011 Nov 21;50(22):11285-7. doi: 10.1021/ic2016066. Epub 2011 Oct 17. Inorg Chem. 2011. PMID: 22004139 Free PMC article.
-
Terminal iron dinitrogen and iron imide complexes supported by a tris(phosphino)borane ligand.Angew Chem Int Ed Engl. 2011 Feb 25;50(9):2063-7. doi: 10.1002/anie.201006918. Epub 2011 Jan 26. Angew Chem Int Ed Engl. 2011. PMID: 21344552 Free PMC article. No abstract available.
-
Conversion of Fe-NH2 to Fe-N2 with release of NH3.J Am Chem Soc. 2013 Jan 16;135(2):534-7. doi: 10.1021/ja307714m. Epub 2013 Jan 8. J Am Chem Soc. 2013. PMID: 23259776 Free PMC article.
-
Proton-Coupled Reduction of an Iron Cyanide Complex to Methane and Ammonia.Angew Chem Int Ed Engl. 2016 Sep 26;55(40):12262-5. doi: 10.1002/anie.201606366. Epub 2016 Sep 8. Angew Chem Int Ed Engl. 2016. PMID: 27607732 Free PMC article.
Cited by
-
Evaluating molecular cobalt complexes for the conversion of N2 to NH3.Inorg Chem. 2015 Oct 5;54(19):9256-62. doi: 10.1021/acs.inorgchem.5b00645. Epub 2015 May 22. Inorg Chem. 2015. PMID: 26001022 Free PMC article.
-
Bonding and stability of dinitrogen-bonded donor base-stabilized Si(0)/Ge(0) species [(cAACMe-Si/Ge)2(N2)]: EDA-NOCV analysis.RSC Adv. 2022 Feb 1;12(7):4081-4093. doi: 10.1039/d1ra07714g. eCollection 2022 Jan 28. RSC Adv. 2022. PMID: 35425464 Free PMC article.
-
EDA-NOCV Calculation for Efficient N2 Binding to the Reduced Ni3S8 Complex: Estimation of Ni-N2 Intrinsic Interaction Energies.ACS Omega. 2021 Dec 2;6(49):33389-33397. doi: 10.1021/acsomega.1c03715. eCollection 2021 Dec 14. ACS Omega. 2021. PMID: 34926888 Free PMC article.
-
Nitrogen Fixation via a Terminal Fe(IV) Nitride.J Am Chem Soc. 2017 Nov 1;139(43):15312-15315. doi: 10.1021/jacs.7b09364. Epub 2017 Oct 19. J Am Chem Soc. 2017. PMID: 28992418 Free PMC article.
-
A Triad of Highly Reduced, Linear Iron Nitrosyl Complexes: {FeNO}(8-10).Angew Chem Int Ed Engl. 2016 Sep 19;55(39):11995-8. doi: 10.1002/anie.201605403. Epub 2016 Aug 25. Angew Chem Int Ed Engl. 2016. PMID: 27560776 Free PMC article.
References
-
- Saouma CT, Müller P, Peters JC. Characterization of structurally unusual diiron NxHy complexes. J Am Chem Soc. 2009;131:10358–10359. - PubMed
-
- Field LD, Li HL, Magill AM. Base-Mediated Conversion of Hydrazine to Diazene and Dinitrogen at an Iron Center. Inorg Chem. 2009;48:5–7. - PubMed
-
- Crossland JL, Balesdent CG, Tyler DR. Intermediates in the reduction of N2 to NH3: synthesis of iron η2 hydrazido(1-) and diazene complexes. Dalton Trans. 2009:4420–4422. - PubMed
-
- Sellmann D, Shaban SY, Heinemann FW. Syntheses, Structures and Reactivity of Electron-Rich Fe and Ru Complexes with the New Pentadentate Ligand Et2NpyS4-H2 {4-(Diethylamino)-2,6-bis[(2-mercaptophenyl)thiomethyl]pyridine} Eur J Inorg Chem. 2004:4591–4601.
-
- Peters JC, Mehn MP. Activation of Small Molecules Ch 3. Wiley-VCH; Weinheim: 2006.
Publication types
MeSH terms
Substances
Associated data
- PubChem-Substance/92708377
- PubChem-Substance/92708378
- PubChem-Substance/92708379
- PubChem-Substance/92708380
- PubChem-Substance/92708381
- PubChem-Substance/92708382
- PubChem-Substance/92708383
- PubChem-Substance/92708384
- PubChem-Substance/92708385
- PubChem-Substance/92708386
- PubChem-Substance/92708387
- PubChem-Substance/92708388
- PubChem-Substance/92708389
- PubChem-Substance/92708390
- PubChem-Substance/92708391
- PubChem-Substance/92708392
- PubChem-Substance/92708393
- PubChem-Substance/92708394
- PubChem-Substance/92708395
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

