Is Nostoc H-NOX a NO sensor or redox switch?
- PMID: 20572679
- PMCID: PMC2914821
- DOI: 10.1021/bi1002234
Is Nostoc H-NOX a NO sensor or redox switch?
Abstract
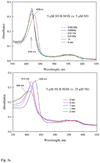
Nostoc sp. (Ns) H-NOX is a heme protein found in symbiotic cyanobacteria, which has approximately 35% sequence identity and high structural homology to the beta subunit of soluble guanylyl cyclase (sGC), suggesting a NO sensing function. However, UV-vis, EPR, NIR MCD, and ligand binding experiments with ferrous and ferric Ns H-NOX indicate significant functional differences between Ns H-NOX and sGC. (1) After NO binding to sGC, the proximal histidine dissociates from the heme iron, causing a conformational change that triggers activation of sGC. In contrast, formation of pentacoordinate (5c) NO heme occurs to only a limited extent in Ns H-NOX, even at >1 mM NO. (2) Unlike sGC, two different hexacoordinate (6c) NO complexes are formed in Ns H-NOX with initial and final absorbance peaks at 418 and 414 nm, and the conversion rate is linearly dependent on [NO], indicating that a second NO binds transiently to catalyze formation of the 414 nm species. (3) sGC is insensitive to oxygen, and ferric sGC prepared by ferricyanide oxidation has a 5c high-spin heme complex. In contrast, Ns H-NOX autoxidizes in 24 h if exposed to air and forms a 6c ferric heme complex, indicating a major conformational change after oxidation and coordination by a second histidine side chain. Such a large conformational transition suggests that Ns H-NOX could function as either a redox or a NO sensor in the cyanobacterium.
Figures















References
-
- Bertini I, Sigel A, Sigel H, editors. Handbooks on metalloproteins. New York: Marcel Dekker, Inc.; 2001.
-
- Kadish KM, Smith KM, Guilard R, editors. The Porphyrin Handbook. Vol. 4, 8, 11. Amsterdam: Academic Press; 2000.
-
- Messerschmidt A, Huber R, Poulos T, Wieghardt K, editors. Handbooks of metalloproteins. Vol. 1. Chichester: John Wiley and Sons, Ltd; 2001.
-
- Gilles-Gonzalez MA, Gonzalez G. Heme-based sensors: defining characteristics, recent developments, and regulatory hypotheses. J Inorg Biochem. 2005;99:1–22. - PubMed
-
- Jain R, Chan MK. Mechanisms of ligand discrimination by heme proteins. J Biol Inorg Chem. 2003;8:1–11. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

