Membrane-associated DNA transport machines
- PMID: 20573715
- PMCID: PMC2890206
- DOI: 10.1101/cshperspect.a000406
Membrane-associated DNA transport machines
Abstract
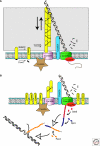
DNA pumps play important roles in bacteria during cell division and during the transfer of genetic material by conjugation and transformation. The FtsK/SpoIIIE proteins carry out the translocation of double-stranded DNA to ensure complete chromosome segregation during cell division. In contrast, the complex molecular machines that mediate conjugation and genetic transformation drive the transport of single stranded DNA. The transformation machine also processes this internalized DNA and mediates its recombination with the resident chromosome during and after uptake, whereas the conjugation apparatus processes DNA before transfer. This article reviews these three types of DNA pumps, with attention to what is understood of their molecular mechanisms, their energetics and their cellular localizations.
Figures



References
-
- Ausmees N, Wahlstedt H, Bagchi S, Elliot MA, Buttner MJ, Flardh K 2007. SmeA, a small membrane protein with multiple functions in Streptomyces sporulation including targeting of a SpoIIIE/FtsK-like protein to cell division septa. Mol Microbiol 65:1458–1473 - PubMed
-
- Aussel L, Barre FX, Aroyo M, Stasiak A, Stasiak AZ, Sherratt D 2002. FtsK Is a DNA motor protein that activates chromosome dimer resolution by switching the catalytic state of the XerC and XerD recombinases. Cell 108:195–205 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
