Optimization of selenoprotein P and other plasma selenium biomarkers for the assessment of the selenium nutritional requirement: a placebo-controlled, double-blind study of selenomethionine supplementation in selenium-deficient Chinese subjects
- PMID: 20573787
- PMCID: PMC2921536
- DOI: 10.3945/ajcn.2010.29642
Optimization of selenoprotein P and other plasma selenium biomarkers for the assessment of the selenium nutritional requirement: a placebo-controlled, double-blind study of selenomethionine supplementation in selenium-deficient Chinese subjects
Abstract
Background: The intake of selenium needed for optimal health has not been established. Selenoproteins perform the functions of selenium, and the selenium intake needed for their full expression is not known.
Objective: This study sought to determine the intake of selenium required to optimize plasma selenoprotein P (SEPP1) and to compare SEPP1 with other plasma selenium biomarkers.
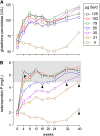
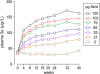
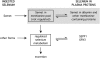
Design: A 40-wk placebo-controlled, double-blind study of selenium repletion was carried out in 98 healthy Chinese subjects who had a daily dietary selenium intake of 14 micro g. Fourteen subjects each were assigned randomly to daily dose groups of 0, 21, 35, 55, 79, 102, and 125 micro g Se as l-selenomethionine. Plasma glutathione peroxidase (GPX) activity, SEPP1, and selenium were measured. A biomarker was considered to be optimized when its value was not different from the mean value of the subjects receiving larger supplements.
Results: The SEPP1 concentration was optimized at 40 wk by the 35- micro g supplement, which indicated that 49 micro g/d could optimize it. GPX activity was optimized by 21 micro g (total ingestion: 35 micro g/d). The selenium concentration showed no tendency to become optimized.
Conclusions: The present results indicate that SEPP1 concentration is the best plasma biomarker studied for assessing optimal expression of all selenoproteins, because its optimization required a larger intake of selenium than did GPX activity. On the basis of the selenium intake needed for SEPP1 optimization with adjustments for body weight and individual variation, ap 75 micro g Se/d as selenomethionine is postulated to allow full expression of selenoproteins in US residents. This trial was registered at clinicaltrials.gov as NCT00428649.
Figures
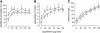
References
-
- Xia Y, Piao J-H, Hill KE, Burk RF. Keshan disease and selenium status of populations in China. : Burk RF, Selenium in biology and human health. New York, NY: Springer Verlag, 1994:182–96
-
- Xia Y. Keshan disease. : Kiple KF, Ornelas KC, The Cambridge world history of food. New York, NY: Cambridge University Press, 2000:939–47
-
- Yang GQ.Keshan Disease: an endemic selenium-related deficiency disease. : Chandra RK, Trace elements in nutrition of children. New York, NY: Vevey/Raven Press, 1985:273–90
-
- Sutter ME, Thomas JD, Brown J, Morgan B. Selenium toxicity: a case of selenosis caused by a nutritional supplement. Ann Intern Med 2008;148:970–1 - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

