Kisspeptin signaling is required for peripheral but not central stimulation of gonadotropin-releasing hormone neurons by NMDA
- PMID: 20573904
- PMCID: PMC6634616
- DOI: 10.1523/JNEUROSCI.5486-09.2010
Kisspeptin signaling is required for peripheral but not central stimulation of gonadotropin-releasing hormone neurons by NMDA
Abstract
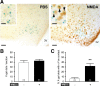
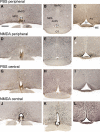
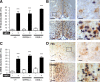
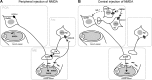
NMDA and kisspeptins can stimulate gonadotropin-releasing hormone (GnRH) release after peripheral or central administration in mice. To determine whether these agonists act independently or through a common pathway, we have examined their ability to stimulate GnRH/luteinizing hormone (LH) release after peripheral or central administration in Kiss1- or Gpr54 (Kiss1r)-null mutant mice. Peripheral injection of NMDA failed to stimulate GnRH/LH release in prepubertal or gonadally intact mutant male mice. Dual-labeling experiments indicated a direct activation of Kiss1-expressing neurons in the arcuate nucleus. In contrast, central injection of NMDA into the lateral ventricle increased plasma LH levels in both Kiss1 and Gpr54 mutant male mice similar to the responses in wild-type mice. Central injection of NMDA stimulated c-Fos expression throughout the hypothalamus but not in GnRH neurons, suggesting an action at the nerve terminals only. In contrast, kisspeptin-10 stimulated LH release after both central and peripheral injection but induced c-Fos expression in GnRH neurons only after central administration. Finally, central injection of NMDA induces c-Fos expression in catecholamine- and nitric oxide-producing neurons in the hypothalamus of mutant mice, indicating a possible kisspeptin-independent GnRH/LH release by NMDA through activation of these neurons. Thus, NMDA may act at both GnRH cell bodies (kisspeptin-independent) and nerve terminals (kisspeptin-dependent) in a dual way to participate in the GnRH/LH secretion in the male mouse.
Figures




Similar articles
-
GnRH(1-5), a metabolite of gonadotropin-releasing hormone, enhances luteinizing hormone release via activation of kisspeptin neurons in female rats.Endocr J. 2020 Apr 28;67(4):409-418. doi: 10.1507/endocrj.EJ19-0444. Epub 2020 Jan 15. Endocr J. 2020. PMID: 31941848
-
Kisspeptin-GPR54 signaling is essential for preovulatory gonadotropin-releasing hormone neuron activation and the luteinizing hormone surge.J Neurosci. 2008 Aug 27;28(35):8691-7. doi: 10.1523/JNEUROSCI.1775-08.2008. J Neurosci. 2008. PMID: 18753370 Free PMC article.
-
The role of kisspeptin-GPR54 signaling in the tonic regulation and surge release of gonadotropin-releasing hormone/luteinizing hormone.J Neurosci. 2007 Oct 31;27(44):12088-95. doi: 10.1523/JNEUROSCI.2748-07.2007. J Neurosci. 2007. PMID: 17978050 Free PMC article.
-
Minireview: kisspeptin neurons as central processors in the regulation of gonadotropin-releasing hormone secretion.Endocrinology. 2006 Mar;147(3):1154-8. doi: 10.1210/en.2005-1282. Epub 2005 Dec 22. Endocrinology. 2006. PMID: 16373418 Review.
-
GPR54 and kisspeptins.Results Probl Cell Differ. 2008;46:117-43. doi: 10.1007/400_2007_050. Results Probl Cell Differ. 2008. PMID: 18193176 Review.
Cited by
-
In vitro and in vivo effects of kisspeptin antagonists p234, p271, p354, and p356 on GPR54 activation.PLoS One. 2017 Jun 26;12(6):e0179156. doi: 10.1371/journal.pone.0179156. eCollection 2017. PLoS One. 2017. PMID: 28650956 Free PMC article.
-
Impact of Altered Lipid Profile in Oral Lichen Planus.Maedica (Bucur). 2023 Mar;18(1):12-18. doi: 10.26574/maedica.2023.18.1.19. Maedica (Bucur). 2023. PMID: 37266475 Free PMC article.
-
Neuroendocrine control by kisspeptins: role in metabolic regulation of fertility.Nat Rev Endocrinol. 2011 Sep 13;8(1):40-53. doi: 10.1038/nrendo.2011.147. Nat Rev Endocrinol. 2011. PMID: 21912400 Review.
-
The ventral premammillary nucleus links metabolic cues and reproduction.Front Endocrinol (Lausanne). 2011 Oct 25;2:57. doi: 10.3389/fendo.2011.00057. eCollection 2011. Front Endocrinol (Lausanne). 2011. PMID: 22649378 Free PMC article.
-
Kisspeptin, gonadotrophin-releasing hormone and oestrogen receptor α colocalise with neuronal nitric oxide synthase neurones in prepubertal female sheep.J Neuroendocrinol. 2018 Jan;30(1):10.1111/jne.12560. doi: 10.1111/jne.12560. J Neuroendocrinol. 2018. PMID: 29178496 Free PMC article.
References
-
- Beauvillain JC, Tramu G. Immunocytochemical demonstration of LH-RH, somatostatin, and ACTH-like peptide in osmium-postfixed, resin-embedded median eminence. J Histochem Cytochem. 1980;28:1014–1017. - PubMed
-
- Bhat GK, Mahesh VB, Lamar CA, Ping L, Aguan K, Brann DW. Histochemical localization of nitric oxide neurons in the hypothalamus: association with gonadotropin-releasing hormone neurons and co-localization with N-methyl-d-aspartate receptors. Neuroendocrinology. 1995;62:187–197. - PubMed
-
- Bourguignon JP, Gerard A, Mathieu J, Simons J, Franchimont P. Pulsatile release of gonadotropin-releasing hormone from hypothalamic explants is restrained by blockade of N-methyl-d,l-aspartate receptors. Endocrinology. 1989;125:1090–1096. - PubMed
-
- Bourguignon JP, Gérard A, Purnelle G, Czajkowski V, Yamanaka C, Lemaître M, Rigo JM, Moonen G, Franchimont P. Duality of glutamatergic and GABAergic control of pulsatile GnRH secretion by rat hypothalamic explants. I. Effects of antisense oligodeoxynucleotides using explants including or excluding the preoptic area. J Neuroendocrinol. 1997;9:183–191. - PubMed
-
- Brace H, Latimer M, Winn P. Neurotoxicity, blood-brain barrier breakdown, demyelination and remyelination associated with NMDA-induced lesions of the rat lateral hypothalamus. Brain Res Bull. 1997;43:447–455. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
