Nerve-released acetylcholine contracts urinary bladder smooth muscle by inducing action potentials independently of IP3-mediated calcium release
- PMID: 20573989
- PMCID: PMC2944423
- DOI: 10.1152/ajpregu.00180.2010
Nerve-released acetylcholine contracts urinary bladder smooth muscle by inducing action potentials independently of IP3-mediated calcium release
Abstract
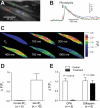
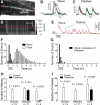
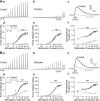
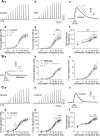
Nerve-released ACh is the main stimulus for contraction of urinary bladder smooth muscle (UBSM). Here, the mechanisms by which ACh contracts UBSM are explored by determining Ca(2+) and electrical signals induced by nerve-released ACh. Photolysis of caged inositol 1,4,5-trisphosphate (IP(3)) evoked Ca(2+) release from the sarcoplasmic reticulum. Electrical field stimulation (20 Hz) induced Ca(2+) waves within the smooth muscle that were present only during stimulus application. Ca(2+) waves were blocked by inhibition of muscarinic ACh receptors (mAChRs) with atropine and depletion of sarcoplasmic reticulum Ca(2+) stores with cyclopiazonic acid (CPA), and therefore likely reflect activation of IP(3) receptors (IP(3)Rs). Electrical field stimulation also increased excitability to induce action potentials (APs) that were accompanied by Ca(2+) flashes, reflecting Ca(2+) entry through voltage-dependent Ca(2+) channels (VDCCs) during the action potential. The evoked Ca(2+) flashes and APs occurred as a burst with a lag time of approximately 1.5 s after onset of stimulation. They were not inhibited by blocking IP(3)-mediated Ca(2+) waves, but by blockers of mAChRs (atropine) and VDCCs (diltiazem). Nerve-evoked contractions of UBSM strips were greatly reduced by blocking VDCCs, but not by preventing IP(3)-mediated Ca(2+) signaling with cyclopiazonic acid or inhibition of PLC with U73122. These results indicate that ACh released from nerve varicosities induces IP(3)-mediated Ca(2+) waves during stimulation; but contrary to expectations, these signals do not appear to participate in contraction. In addition, our data provide compelling evidence that UBSM contractions evoked by nerve-released ACh depend on increased excitability and the resultant Ca(2+) entry through VDCCs during APs.
Figures


Similar articles
-
Nerve-evoked purinergic signalling suppresses action potentials, Ca2+ flashes and contractility evoked by muscarinic receptor activation in mouse urinary bladder smooth muscle.J Physiol. 2009 Nov 1;587(Pt 21):5275-88. doi: 10.1113/jphysiol.2009.178806. Epub 2009 Sep 7. J Physiol. 2009. PMID: 19736301 Free PMC article.
-
Negative feedback regulation of nerve-mediated contractions by KCa channels in mouse urinary bladder smooth muscle.Am J Physiol Regul Integr Comp Physiol. 2005 Aug;289(2):R402-R409. doi: 10.1152/ajpregu.00488.2004. Epub 2005 Apr 21. Am J Physiol Regul Integr Comp Physiol. 2005. PMID: 15845880
-
Spontaneous electrical rhythmicity and the role of the sarcoplasmic reticulum in the excitability of guinea pig gallbladder smooth muscle cells.Am J Physiol Gastrointest Liver Physiol. 2006 Apr;290(4):G655-64. doi: 10.1152/ajpgi.00310.2005. Epub 2005 Nov 17. Am J Physiol Gastrointest Liver Physiol. 2006. PMID: 16293655
-
Sarcoplasmic reticulum and membrane currents.Novartis Found Symp. 2002;246:189-203; discussion 203-7, 221-7. Novartis Found Symp. 2002. PMID: 12164309 Review.
-
Calcium signaling in smooth muscle.Cold Spring Harb Perspect Biol. 2011 Sep 1;3(9):a004549. doi: 10.1101/cshperspect.a004549. Cold Spring Harb Perspect Biol. 2011. PMID: 21709182 Free PMC article. Review.
Cited by
-
Local coupling of TRPC6 to ANO1/TMEM16A channels in smooth muscle cells amplifies vasoconstriction in cerebral arteries.Am J Physiol Cell Physiol. 2016 Jun 1;310(11):C1001-9. doi: 10.1152/ajpcell.00092.2016. Epub 2016 May 4. Am J Physiol Cell Physiol. 2016. PMID: 27147559 Free PMC article.
-
Plastic induced urinary tract disease and dysfunction: a scoping review.J Expo Sci Environ Epidemiol. 2024 Aug 31. doi: 10.1038/s41370-024-00709-3. Online ahead of print. J Expo Sci Environ Epidemiol. 2024. PMID: 39217203 Review.
-
Calcium Signaling Pathways: Key Pathways in the Regulation of Obesity.Int J Mol Sci. 2019 Jun 5;20(11):2768. doi: 10.3390/ijms20112768. Int J Mol Sci. 2019. PMID: 31195699 Free PMC article. Review.
-
NS19504: a novel BK channel activator with relaxing effect on bladder smooth muscle spontaneous phasic contractions.J Pharmacol Exp Ther. 2014 Sep;350(3):520-30. doi: 10.1124/jpet.113.212662. Epub 2014 Jun 20. J Pharmacol Exp Ther. 2014. PMID: 24951278 Free PMC article.
-
Marine Sponges as a Drug Treasure.Biomol Ther (Seoul). 2016 Jul 1;24(4):347-62. doi: 10.4062/biomolther.2016.067. Biomol Ther (Seoul). 2016. PMID: 27350338 Free PMC article. Review.
References
-
- Andersson KE, Arner A. Urinary bladder contraction and relaxation: physiology and pathophysiology. Physiol Rev 84: 935–986, 2004 - PubMed
-
- Bonev AD, Nelson MT. Muscarinic inhibition of ATP-sensitive K+ channels by protein kinase C in urinary bladder smooth muscle. Am J Physiol Cell Physiol 265: C1723–C1728, 1993 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

