Simple screening method for autoantigen proteins using the N-terminal biotinylated protein library produced by wheat cell-free synthesis
- PMID: 20575507
- PMCID: PMC2917173
- DOI: 10.1021/pr9010553
Simple screening method for autoantigen proteins using the N-terminal biotinylated protein library produced by wheat cell-free synthesis
Abstract
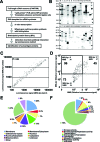
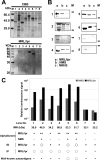
Autoimmune diseases are a heterogeneous group of diseases characterized by immune reactions against either a major or a limited number of the bodies own autoantigens, causing inflammation and damage to tissues and organs. Thus, identification of autoantigens is an important first step to understanding autoimmune diseases. Here we demonstrate a simple screening method for identification of autoantigens reacting with patient serum antibodies by combination of an N-terminal biotinylated protein library (BPL), produced using a wheat cell-free protein production system, and a commercially available luminescence system. Optimization studies using well-characterized autoantigens showed specific interactions between N-terminal biotinylated proteins and antibody that were sensitively detected under homogeneous reaction conditions. In this optimized assay, 1 microL of the translation mixture expressing the biotinylated proteins produced significant luminescence signal by addition of diluted serum between 1:500 and 1:10 000 in 25 microL of reaction volume. For the BPL construction, 214 mouse genes, consisting of 103 well-known autoantigens and 111 genes in the mouse autoimmune susceptibility loci, and the sera of MRL/lpr mouse were used as an autoimmune model. By this screening method, 25 well-known autoantigens and 71 proteins in the loci were identified as autoantigen proteins specifically reacting with sera antibodies. Cross-referencing with the Gene Ontology Database, 26 and 38 of autoantigen proteins were predicted to have nuclear localization and identified as membrane and/or extracellular proteins. The immune reaction of six randomly selected proteins was confirmed by immunoprecipitation and/or immunoblot analyses. Interestingly, three autoantigen proteins were recognized by immunoprecipitation but not by immunoblot analysis. These results suggest that the BPL-based method could provide a simple system for screening of autoantigen proteins and would help with identification of autoantigen proteins reacting with antibodies that recognize folded proteins, rather than denatured or unfolded forms.
Figures


Similar articles
-
Novel approach to identifying autoantibodies in rheumatoid synovitis with a biotinylated human autoantigen library and the enzyme-labeled antigen method.J Immunol Methods. 2013 Jan 31;387(1-2):57-70. doi: 10.1016/j.jim.2012.09.011. Epub 2012 Oct 5. J Immunol Methods. 2013. PMID: 23044167
-
Identification of TPIT and other novel autoantigens in lymphocytic hypophysitis: immunoscreening of a pituitary cDNA library and development of immunoprecipitation assays.Eur J Endocrinol. 2012 Mar;166(3):391-8. doi: 10.1530/EJE-11-1015. Epub 2011 Dec 22. Eur J Endocrinol. 2012. PMID: 22193973 Free PMC article.
-
Autoantigen discovery with a synthetic human peptidome.Nat Biotechnol. 2011 May 22;29(6):535-41. doi: 10.1038/nbt.1856. Nat Biotechnol. 2011. PMID: 21602805 Free PMC article.
-
Autoantigenomics: Holistic characterization of autoantigen repertoires for a better understanding of autoimmune diseases.Autoimmun Rev. 2020 Feb;19(2):102450. doi: 10.1016/j.autrev.2019.102450. Epub 2019 Dec 12. Autoimmun Rev. 2020. PMID: 31838165 Review.
-
Autoantigen complementarity: a new theory implicating complementary proteins as initiators of autoimmune disease.J Mol Med (Berl). 2005 Jan;83(1):12-25. doi: 10.1007/s00109-004-0615-3. Epub 2004 Dec 11. J Mol Med (Berl). 2005. PMID: 15592920 Review.
Cited by
-
Specific in situ visualization of plasma cells producing antibodies against Porphyromonas gingivalis in gingival radicular cyst: application of the enzyme-labeled antigen method.J Histochem Cytochem. 2011 Jul;59(7):673-89. doi: 10.1369/0022155411408906. Epub 2011 Apr 27. J Histochem Cytochem. 2011. PMID: 21525188 Free PMC article.
-
Rapid mapping of interactions between Human SNX-BAR proteins measured in vitro by AlphaScreen and single-molecule spectroscopy.Mol Cell Proteomics. 2014 Sep;13(9):2233-45. doi: 10.1074/mcp.M113.037275. Epub 2014 May 27. Mol Cell Proteomics. 2014. PMID: 24866125 Free PMC article.
-
H11/HSPB8 Restricts HIV-2 Vpx to Restore the Anti-Viral Activity of SAMHD1.Front Microbiol. 2016 Jun 13;7:883. doi: 10.3389/fmicb.2016.00883. eCollection 2016. Front Microbiol. 2016. PMID: 27379031 Free PMC article.
-
Easy Synthesis of Complex Biomolecular Assemblies: Wheat Germ Cell-Free Protein Expression in Structural Biology.Front Mol Biosci. 2021 Mar 25;8:639587. doi: 10.3389/fmolb.2021.639587. eCollection 2021. Front Mol Biosci. 2021. PMID: 33842544 Free PMC article. Review.
-
PfMSA180 is a novel Plasmodium falciparum vaccine antigen that interacts with human erythrocyte integrin associated protein (CD47).Sci Rep. 2019 Apr 11;9(1):5923. doi: 10.1038/s41598-019-42366-9. Sci Rep. 2019. PMID: 30976034 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

