Sodium 4-phenylbutyrate ameliorates the effects of cataract-causing mutant gammaD-crystallin in cultured cells
- PMID: 20577655
- PMCID: PMC2890554
Sodium 4-phenylbutyrate ameliorates the effects of cataract-causing mutant gammaD-crystallin in cultured cells
Abstract
Purpose: gammaD-Crystallin (CRYGD) is a major structural lens crystallin and its mutations result in congenital cataract formation. In this study, we attempted to correct the altered protein features of G165fsX8 CRYGD protein with small chemical molecules.
Methods: Recombinant FLAG-tagged mutants (R15C, R15S, P24T, R61C, and G165fsX8) of CRYGD were expressed in COS-7 cells and treated with small chemical molecules with reported protein chaperoning properties (sodium 4-phenylbutyrate [4-PBA], trimethylamine N-oxide [TMAO], and glycerol and DMSO [DMSO]). Protein solubility in 0.5% Triton X-100 and subcellular distribution was examined by western blotting and immunofluorescence, respectively. Apoptosis was assayed as the percentage of fragmented nuclei in transfected cells. Expression of heat-shock proteins (Hsp70 and Hsp90) was examined by reverse transcription-polymerase chain reaction analysis.
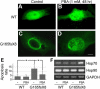
Results: Unlike WT and most mutants (R15C, R15S, P24T, and R61C) of CRYGD, G165fsX8 CRYGD was significantly insoluble in 0.5% Triton X-100. This insolubility was alleviated by dose-dependent 4-PBA treatment. The treatment relieved the mislocalization of G165fsX8 CRYGD from the nuclear envelope. Also, 4-PBA treatment reduced cell apoptosis and caused an upregulation of Hsp70.
Conclusions: 4-PBA treatment reduced the defective phenotype of mutant G165fsX8 CRYGD and rescued the affected cells from apoptosis. This could be a potential treatment for lens structural protein and prevent lens opacity in cataract formation.
Figures
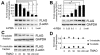

Similar articles
-
A novel gammaD-crystallin mutation causes mild changes in protein properties but leads to congenital coralliform cataract.Mol Vis. 2009 Aug 6;15:1521-9. Mol Vis. 2009. PMID: 19668596 Free PMC article.
-
A novel deletion variant of gammaD-crystallin responsible for congenital nuclear cataract.Mol Vis. 2007 Nov 7;13:2096-104. Mol Vis. 2007. PMID: 18079686
-
Trimethylamine N-oxide alleviates the severe aggregation and ER stress caused by G98R alphaA-crystallin.Mol Vis. 2009 Dec 19;15:2829-40. Mol Vis. 2009. PMID: 20029648 Free PMC article.
-
A potential novel role of the R36P mutation in CRYGD in congenial cataract.Mol Vis. 2024 Jun 26;30:260-267. eCollection 2024. Mol Vis. 2024. PMID: 39563676 Free PMC article.
-
Mutation analysis of CRYAA, CRYGC, and CRYGD associated with autosomal dominant congenital cataract in Brazilian families.Mol Vis. 2009;15:793-800. Epub 2009 Apr 17. Mol Vis. 2009. PMID: 19390652 Free PMC article.
Cited by
-
Patterns of gene expression in microarrays and expressed sequence tags from normal and cataractous lenses.Hum Genomics. 2012 Sep 1;6(1):14. doi: 10.1186/1479-7364-6-14. Hum Genomics. 2012. PMID: 23244575 Free PMC article.
-
Topical ocular sodium 4-phenylbutyrate rescues glaucoma in a myocilin mouse model of primary open-angle glaucoma.Invest Ophthalmol Vis Sci. 2012 Mar 21;53(3):1557-65. doi: 10.1167/iovs.11-8837. Print 2012 Mar. Invest Ophthalmol Vis Sci. 2012. PMID: 22328638 Free PMC article.
-
Discovery of Molecular Therapeutics for Glaucoma: Challenges, Successes, and Promising Directions.J Med Chem. 2016 Feb 11;59(3):788-809. doi: 10.1021/acs.jmedchem.5b00828. Epub 2015 Sep 25. J Med Chem. 2016. PMID: 26356532 Free PMC article. Review.
-
Targeting cellular stress in vitro improves osteoblast homeostasis, matrix collagen content and mineralization in two murine models of osteogenesis imperfecta.Matrix Biol. 2021 Apr;98:1-20. doi: 10.1016/j.matbio.2021.03.001. Epub 2021 Mar 31. Matrix Biol. 2021. PMID: 33798677 Free PMC article.
-
Chemical chaperone treatment improves levels and distributions of connexins in Cx50D47A mouse lenses.Exp Eye Res. 2018 Oct;175:192-198. doi: 10.1016/j.exer.2018.06.015. Epub 2018 Jun 18. Exp Eye Res. 2018. PMID: 29913165 Free PMC article.
References
-
- Aridor M, Hannan LA. Traffic jams II: an update of diseases of intracellular transport. Traffic. 2002;3:781–90. - PubMed
-
- Broadley SA, Hartl FU. The role of molecular chaperones in human misfolding diseases. FEBS Lett. 2009;583:2647–53. - PubMed
-
- Trombetta ES, Parodi AJ. Quality control and protein folding in the secretory pathway. Annu Rev Cell Dev Biol. 2003;19:649–76. - PubMed
-
- Ellgaard L, Molinari M, Helenius A. Setting the standards: quality control in the secretory pathway. Science. 1999;286:1882–8. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
