Discovery and rediscoveries of Golgi cells
- PMID: 20581044
- PMCID: PMC2998217
- DOI: 10.1113/jphysiol.2010.189605
Discovery and rediscoveries of Golgi cells
Abstract
When Camillo Golgi invented the black reaction in 1873 and first described the fine anatomical structure of the nervous system, he described a ‘big nerve cell’ that later took his name, the Golgi cell of cerebellum (‘Golgi’schen Zellen’, Gustaf Retzius, 1892). The Golgi cell was then proposed as the prototype of type-II interneurons, which form complex connections and exert their actions exclusively within the local network. Santiago Ramón y Cajal (who received the Nobel Prize with Golgi in 1906) proceeded to a detailed description of Golgi cell morphological characteristics, but functional insight remained very limited for many years. The first rediscovery happened in the 1960s, when neurophysiological analysis in vivo revealed that Golgi cells are inhibitory interneurons. This finding promoted the development of two major cerebellar theories, the ‘beam theory’ of John Eccles and the ‘motor learning theory’ of David Marr, in which the Golgi cells regulate the spatial organisation and the gain of input signals to be processed and learned by the cerebellar circuit. However, the matter was not set and a series of pioneering observations using single unit recordings and electronmicroscopy raised new issues that could not be fully explored until the 1990s. Then, the advent of new electrophysiological and imaging techniques in vitro and in vivo demonstrated the cellular and network activities of these neurons. Now we know that Golgi cells, through complex systems of chemical and electrical synapses, effectively control the spatio-temporal organisation of cerebellar responses. The Golgi cells regulate the timing and number of spikes emitted by granule cells and coordinate their coherent activity. Moreover, the Golgi cells regulate the induction of long-term synaptic plasticity along the mossy fibre pathway. Eventually, the Golgi cells transform the granular layer of cerebellum into an adaptable spatio-temporal filter capable of performing several kinds of logical operation. After more than a century, Golgi’s intuition that the Golgi cell had to generate under a new perspective complex ensemble effects at the network level has finally been demonstrated.
Figures



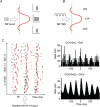

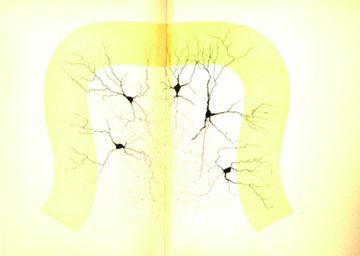

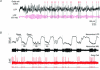
Similar articles
-
Synchronization of golgi and granule cell firing in a detailed network model of the cerebellar granule cell layer.J Neurophysiol. 1998 Nov;80(5):2521-37. doi: 10.1152/jn.1998.80.5.2521. J Neurophysiol. 1998. PMID: 9819260
-
The diffuse nervous network of Camillo Golgi: facts and fiction.Brain Res Rev. 2011 Jan 7;66(1-2):75-82. doi: 10.1016/j.brainresrev.2010.09.005. Epub 2010 Sep 16. Brain Res Rev. 2011. PMID: 20840856
-
Cerebellar circuitry as a neuronal machine.Prog Neurobiol. 2006 Feb-Apr;78(3-5):272-303. doi: 10.1016/j.pneurobio.2006.02.006. Prog Neurobiol. 2006. PMID: 16759785 Review.
-
How the 1906 Nobel Prize in Physiology or Medicine was shared between Golgi and Cajal.Brain Res Rev. 2007 Oct;55(2):490-8. doi: 10.1016/j.brainresrev.2006.11.004. Epub 2007 Jan 9. Brain Res Rev. 2007. PMID: 17306375
-
The cerebellar network: from structure to function and dynamics.Brain Res Rev. 2011 Jan 7;66(1-2):5-15. doi: 10.1016/j.brainresrev.2010.10.002. Epub 2010 Oct 13. Brain Res Rev. 2011. PMID: 20950649 Review.
Cited by
-
Robustness effect of gap junctions between Golgi cells on cerebellar cortex oscillations.Neural Syst Circuits. 2011 Mar 1;1(1):7. doi: 10.1186/2042-1001-1-7. Neural Syst Circuits. 2011. PMID: 22330240 Free PMC article.
-
Golgi cell activity during eyeblink conditioning in decerebrate ferrets.Cerebellum. 2014 Feb;13(1):42-5. doi: 10.1007/s12311-013-0518-3. Cerebellum. 2014. PMID: 23982588
-
Computational Theory Underlying Acute Vestibulo-ocular Reflex Motor Learning with Cerebellar Long-Term Depression and Long-Term Potentiation.Cerebellum. 2017 Aug;16(4):827-839. doi: 10.1007/s12311-017-0857-6. Cerebellum. 2017. PMID: 28444617
-
The cerebellum converts input data into a hyper low-resolution granule cell code with spatial dimensions: a hypothesis.R Soc Open Sci. 2025 Mar 26;12(3):241665. doi: 10.1098/rsos.241665. eCollection 2025 Mar. R Soc Open Sci. 2025. PMID: 40144291 Free PMC article.
-
Model cerebellar granule cells can faithfully transmit modulated firing rate signals.Front Cell Neurosci. 2014 Oct 13;8:304. doi: 10.3389/fncel.2014.00304. eCollection 2014. Front Cell Neurosci. 2014. PMID: 25352777 Free PMC article.
References
-
- Albus JS. A theory of cerebellar function. Math Biosci. 1971;10:25–61.
-
- Ambrosi G, Flace P, Lorusso L, Girolamo F, Rizzi A, Bosco L, Errede M, Virgintino D, Roncali L, Benagiano V. Non-traditional large neurons in the granular layer of the cerebellar cortex. Eur J Histochem. 2007;51(Suppl 1):59–64. - PubMed
-
- Apps R, Hawkes R. Cerebellar cortical organization: a one-map hypothesis. Nat Rev Neurosci. 2009;10:670–681. - PubMed
-
- Bower JM. Is the cerebellum sensory for motor's sake, or motor for sensory's sake: the view from the whiskers of a rat? Prog Brain Res. 1997;114:463–96. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

