VERDANDI is a direct target of the MADS domain ovule identity complex and affects embryo sac differentiation in Arabidopsis
- PMID: 20581305
- PMCID: PMC2910977
- DOI: 10.1105/tpc.109.068627
VERDANDI is a direct target of the MADS domain ovule identity complex and affects embryo sac differentiation in Arabidopsis
Abstract
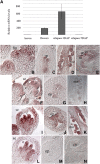
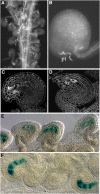
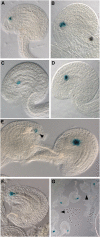
In Arabidopsis thaliana, the three MADS box genes SEEDSTICK (STK), SHATTERPROOF1 (SHP1), and SHP2 redundantly regulate ovule development. Protein interaction studies have shown that a multimeric complex composed of the ovule identity proteins together with the SEPALLATA MADS domain proteins is necessary to determine ovule identity. Despite the extensive knowledge that has become available about these MADS domain transcription factors, little is known regarding the genes that they regulate. Here, we show that STK, SHP1, and SHP2 redundantly regulate VERDANDI (VDD), a putative transcription factor that belongs to the plant-specific B3 superfamily. The vdd mutant shows defects during the fertilization process resulting in semisterility. Analysis of the vdd mutant female gametophytes indicates that antipodal and synergid cell identity and/or differentiation are affected. Our results provide insights into the pathways regulated by the ovule identity factors and the role of the downstream target gene VDD in female gametophyte development.
Figures


References
-
- Battaglia R., Brambilla V., Colombo L. (2008). Morphological analysis of female gametophyte development in the bel1 stk shp1 shp2 mutant. Plant Biosyst. 142: 643–649
-
- Battaglia R., Brambilla V., Colombo L., Stuitje A.R., Kater M.M. (2006). Functional analysis of MADS-box protein complexes controlling ovule development in Arabidopsis using the ethanol-inducible alc gene-expression system. Mech. Dev. 123: 267–276 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

