Knockouts of SOD1 and GPX1 exert different impacts on murine islet function and pancreatic integrity
- PMID: 20586612
- PMCID: PMC3026657
- DOI: 10.1089/ars.2010.3302
Knockouts of SOD1 and GPX1 exert different impacts on murine islet function and pancreatic integrity
Abstract
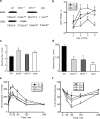
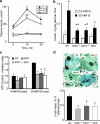
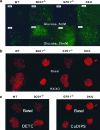
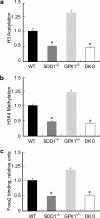
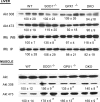
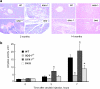
Metabolic subtlety and clinical relevance of different forms of reactive oxygen species in diabetes remain unclear. Using single knockout of Cu,Zn-superoxide dismutase (SOD1(-/-)) or Se-glutathione peroxidase-1 (GPX1(-/-)) and their double-knockout (DKO) mouse models, we determined if elevating endogenously-derived superoxide and hydroperoxide exerted distinct impacts and mechanisms on body glucose homeostasis. Whereas the three knockout groups displayed decreased plasma insulin concentrations and islet β-cells mass, only SOD1(-/-) showed decreased body weight, increased blood glucose, and blocked glucose-stimulated insulin secretion. Null of SOD1 and GPX1 elevated respective islet superoxide and hydroperoxide production, and upregulated p53 phosphorylation. Knockout of SOD1 downregulated the foxhead box A2/pancreatic and duodenal homeobox 1 pathway in a superoxide-dependent fashion at epigenetic, mRNA, and protein levels in islets, but improved insulin signaling in liver and muscle. The SOD1(-/-) mice showed more apparent pancreatitis than the GPX1(-/-) mice that were more susceptible to the cerulein-induced amylase increase. Knockout of SOD1 impaired islet function, pancreas integrity, and body glucose homeostasis more than that of GPX1. Simultaneous ablation of both enzymes did not result in additive or aggravated metabolic outcomes.
Figures


Similar articles
-
Two tales of antioxidant enzymes on β cells and diabetes.Antioxid Redox Signal. 2011 Feb 1;14(3):489-503. doi: 10.1089/ars.2010.3416. Epub 2010 Oct 19. Antioxid Redox Signal. 2011. PMID: 20618069 Free PMC article. Review.
-
Glutathione peroxidase mimic ebselen improves glucose-stimulated insulin secretion in murine islets.Antioxid Redox Signal. 2014 Jan 10;20(2):191-203. doi: 10.1089/ars.2013.5361. Epub 2013 Aug 9. Antioxid Redox Signal. 2014. PMID: 23795780 Free PMC article.
-
Impact of Cu, Zn-superoxide dismutase and Se-dependent glutathione peroxidase-1 knockouts on acetaminophen-induced cell death and related signaling in murine liver.Exp Biol Med (Maywood). 2006 Dec;231(11):1726-32. doi: 10.1177/153537020623101109. Exp Biol Med (Maywood). 2006. PMID: 17138759
-
Knockout of SOD1 alters murine hepatic glycolysis, gluconeogenesis, and lipogenesis.Free Radic Biol Med. 2012 Nov 1;53(9):1689-96. doi: 10.1016/j.freeradbiomed.2012.08.570. Epub 2012 Aug 25. Free Radic Biol Med. 2012. PMID: 22974764 Free PMC article.
-
Interference of selenium and selenoproteins with the insulin-regulated carbohydrate and lipid metabolism.Free Radic Biol Med. 2013 Dec;65:1538-1547. doi: 10.1016/j.freeradbiomed.2013.07.016. Epub 2013 Jul 18. Free Radic Biol Med. 2013. PMID: 23872396 Review.
Cited by
-
Selenium and diabetes--evidence from animal studies.Free Radic Biol Med. 2013 Dec;65:1548-1556. doi: 10.1016/j.freeradbiomed.2013.07.012. Epub 2013 Jul 16. Free Radic Biol Med. 2013. PMID: 23867154 Free PMC article. Review.
-
GSH levels affect weight loss in individuals with metabolic syndrome and obesity following dietary therapy.Exp Ther Med. 2018 Aug;16(2):635-642. doi: 10.3892/etm.2018.6204. Epub 2018 May 23. Exp Ther Med. 2018. PMID: 30116319 Free PMC article.
-
Secondary analysis of publicly available data reveals superoxide and oxygen radical pathways are enriched for associations between type 2 diabetes and low-frequency variants.Ann Hum Genet. 2013 Nov;77(6):472-81. doi: 10.1111/ahg.12035. Epub 2013 Aug 14. Ann Hum Genet. 2013. PMID: 23941231 Free PMC article.
-
Redox regulation of genome stability by effects on gene expression, epigenetic pathways and DNA damage/repair.Redox Biol. 2015 Aug;5:275-289. doi: 10.1016/j.redox.2015.05.008. Epub 2015 Jun 3. Redox Biol. 2015. PMID: 26079210 Free PMC article. Review.
-
Pancreatic β-cell dysfunction in type 2 diabetes: Implications of inflammation and oxidative stress.World J Diabetes. 2023 Mar 15;14(3):130-146. doi: 10.4239/wjd.v14.i3.130. World J Diabetes. 2023. PMID: 37035220 Free PMC article. Review.
References
-
- Adachi T. Inoue M. Hara H. Maehata E. Suzuki S. Relationship of plasma extracellular-superoxide dismutase level with insulin resistance in type 2 diabetic patients. J Endocrinol. 2004;181:413–417. - PubMed
-
- Ahren B. Islet G protein-coupled receptors as potential targets for treatment of type 2 diabetes. Nat Rev Drug Discov. 2009;8:369–385. - PubMed
-
- Aylon Y. Oren M. Living with p53, dying of p53. Cell. 2007;130:597–600. - PubMed
-
- Bernard C. Berthault M-F. Saulnier C. Ktorza A. Neogenesis vs. apoptosis as main components of pancreatic ß cell mass changes in glucose-infused normal and mildly diabetic adult rats. FASEB J. 1999;13:1195–1205. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

