Defective translocation of PKCepsilon in EtOH-induced inhibition of Mg2+ accumulation in rat hepatocytes
- PMID: 20586749
- PMCID: PMC2929311
- DOI: 10.1111/j.1530-0277.2010.01252.x
Defective translocation of PKCepsilon in EtOH-induced inhibition of Mg2+ accumulation in rat hepatocytes
Abstract
Background: Rats chronically fed ethanol for 3 weeks presented a marked decreased in total hepatic Mg(2+) content and required approximately 12 days to restore Mg(2+) homeostasis upon ethanol withdrawal. This study was aimed at investigating the mechanisms responsible for the EtOH-induced delay.
Methods: Hepatocytes from rats fed ethanol for 3 weeks (Lieber-De Carli diet-chronic model), rats re-fed a control diet for varying periods of time following ethanol withdrawal, and age-matched control rats fed a liquid or a pellet diet were used. As acute models, hepatocytes from control animals or HepG2 cells were exposed to varying doses of ethanol in vitro for 8 minutes.
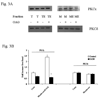
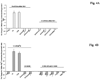
Results: Hepatocytes from ethanol-fed rats presented a marked inhibition of Mg(2+) accumulation and a defective translocation of PKCepsilon to the cell membrane. Upon ethanol withdrawal, 12 days were necessary for PKCepsilon translocation and Mg(2+) accumulation to return to normal levels. Exposure of control hepatocytes or HepG2 cells to a dose of ethanol as low as 0.01% for 8 minutes was already sufficient to inhibit Mg(2+) accumulation and PKCepsilon translocation for more than 60 minutes. Also in this model, recovery of Mg(2+) accumulation was associated with restoration of PKCepsilon translocation. The use of specific antisense in HepG2 cells confirmed the involvement of PKCepsilon in modulating Mg(2+) accumulation.
Conclusions: Translocation of PKCepsilon isoform to the hepatocyte membrane is essential for Mg(2+) accumulation to occur. Both acute and chronic ethanol administrations inhibit Mg(2+) accumulation by specifically altering PKCepsilon translocation to the cell membrane.
Figures








Similar articles
-
Delayed restoration of Mg2+ content and transport in liver cells following ethanol withdrawal.Am J Physiol Gastrointest Liver Physiol. 2009 Oct;297(4):G621-31. doi: 10.1152/ajpgi.90652.2008. Epub 2009 Jun 25. Am J Physiol Gastrointest Liver Physiol. 2009. PMID: 19556361 Free PMC article.
-
Ethanol induces apoptosis in hepatocytes by a pathway involving novel protein kinase C isoforms.Cell Signal. 2007 Nov;19(11):2339-50. doi: 10.1016/j.cellsig.2007.07.013. Epub 2007 Jul 27. Cell Signal. 2007. PMID: 17728104
-
Chronic EtOH administration alters liver Mg2+ homeostasis.Am J Physiol Gastrointest Liver Physiol. 2003 Jan;284(1):G57-67. doi: 10.1152/ajpgi.00153.2002. Am J Physiol Gastrointest Liver Physiol. 2003. PMID: 12488234
-
PKCε contributes to chronic ethanol-induced steatosis in mice but not inflammation and necrosis.Alcohol Clin Exp Res. 2014 Mar;38(3):801-9. doi: 10.1111/acer.12324. Epub 2014 Jan 31. Alcohol Clin Exp Res. 2014. PMID: 24483773 Free PMC article.
-
Mechanism of inhibition of taurolithocholate-induced retrieval of plasma membrane MRP2 by cyclic AMP and tauroursodeoxycholate.Physiol Rep. 2017 Dec;5(23):e13529. doi: 10.14814/phy2.13529. Physiol Rep. 2017. PMID: 29192063 Free PMC article.
Cited by
-
Effect of Alcohol Administration on Mg2+ Homeostasis in H9C2 Cells.J Cardiovasc Dis Diagn. 2014 Oct 23;2(6):179. doi: 10.4172/2329-9517.1000179. J Cardiovasc Dis Diagn. 2014. PMID: 25793216 Free PMC article.
-
Cellular magnesium homeostasis.Arch Biochem Biophys. 2011 Aug 1;512(1):1-23. doi: 10.1016/j.abb.2011.05.010. Epub 2011 May 27. Arch Biochem Biophys. 2011. PMID: 21640700 Free PMC article. Review.
-
Reduced cellular Mg²⁺ content enhances hexose 6-phosphate dehydrogenase activity and expression in HepG2 and HL-60 cells.Arch Biochem Biophys. 2014 Apr 15;548:11-9. doi: 10.1016/j.abb.2014.02.016. Epub 2014 Mar 11. Arch Biochem Biophys. 2014. PMID: 24631573 Free PMC article.
-
Effect of acute and prolonged alcohol administration on Mg(2+) homeostasis in cardiac cells.Alcohol. 2015 May;49(3):265-73. doi: 10.1016/j.alcohol.2015.02.002. Epub 2015 Mar 4. Alcohol. 2015. PMID: 25800156 Free PMC article.
-
Enhanced glucose 6-phosphatase activity in liver of rats exposed to Mg(2+)-deficient diet.Arch Biochem Biophys. 2011 May 15;509(2):157-63. doi: 10.1016/j.abb.2011.03.003. Epub 2011 Mar 21. Arch Biochem Biophys. 2011. PMID: 21402051 Free PMC article.
References
-
- Cao G, Thebault S, van der Wijst J, van der Kemp A-M, Lasonder E, Bindels RJM. RACK1 inhibits TRPM6 activity via phosphorylation of the fused alpha-kinase domain. Curr Biol. 2008;18:168–178. - PubMed
-
- Cefaratti C, Romani A. Functional characterization of two distinct Mg2+ extrusion mechanisms in cardiac sarcolemmal vesicles. Mol Cell Biochem. 2007;303:63–72. - PubMed
-
- Csermely P, Fodor P, Somogyi J. The tumor promoter tetradecanoylphorbol-13 acetate elicits the redistribution of heavy metal in subcellular fractions of rabbit thymocytes as measured by plasma emission spectroscopy. Carcinogenesis. 1987;8:1663–1666. - PubMed
-
- Díaz-Guerra MJ, Junco M, Boscá L. Oleic acid promotes changes in the subcellular distribution of protein kinase C in isolated hepatocytes. J Biol Chem. 1991;266:23568–23576. - PubMed
-
- Domenicotti C, Paola D, Lamedica A, Ricciarelli R, Chiarpotto E, Marinari UM, Poli G, Melloni E, Pronzato MA. Effects of ethanol metabolism on PKC activity in isolated rat hepatocytes. Chem Biol Interact. 1996;100:155–163. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

