Yeast karyopherin Kap95 is required for cell cycle progression at Start
- PMID: 20587033
- PMCID: PMC2904269
- DOI: 10.1186/1471-2121-11-47
Yeast karyopherin Kap95 is required for cell cycle progression at Start
Abstract
Background: The control of the subcellular localization of cell cycle regulators has emerged as a crucial mechanism in cell division regulation. The active transport of proteins between the nucleus and the cytoplasm is mediated by the transport receptors of the beta-karyopherin family. In this work we characterized the terminal phenotype of a mutant strain in beta-karyopherin Kap95, a component of the classical nuclear import pathway.
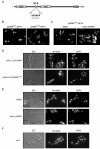
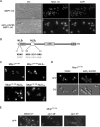
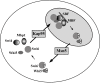
Results: When KAP95 was inactivated, most cells arrested at the G2/M phase of the cell cycle, which is in agreement with the results observed in mutants in the other components of this pathway. However, a number of cells accumulate at G1, suggesting a novel role of Kap95 and the classical import pathway at Start. We investigated the localization of Start transcription factors. It is known that Swi6 contains a classical NLS that interacts with importin alpha. Here we show that the in vivo nuclear import of Swi6 depends on Kap95. For Swi4, we identified a functional NLS between amino acids 371 and 376 that is sufficient and necessary for Swi4 to enter the nucleus. The nuclear import driven by this NLS is mediated by karyopherins Kap95 and Srp1. Inactivation of Kap95 also produces a dramatic change in the localization of Mbp1 since the protein is mainly detected in the cytoplasm. Two functionally redundant Kap95- and Srp1-dependent NLSs were identified in Mbp1 between amino acids 27-30 and 166-181. Nuclear accumulation was not completely abolished in a kap95 mutant or in the Mbp1 mutated in the two NLSs, suggesting that alternative pathways might contribute to the Mbp1 nuclear import to a lesser extent.
Conclusions: Kap95 plays an essential role at the initiation of the cell cycle by driving the nuclear import of Swi4, Swi6 and Mbp1, the three transcription factors responsible for the gene expression at Start. This transport depends on the specific nuclear localization signals present in cargo proteins.
Figures



Similar articles
-
Nuclear import of the yeast hexokinase 2 protein requires α/β-importin-dependent pathway.J Biol Chem. 2012 Jan 27;287(5):3518-29. doi: 10.1074/jbc.M111.317230. Epub 2011 Dec 7. J Biol Chem. 2012. PMID: 22157003 Free PMC article.
-
Yeast importin-β is required for nuclear import of the Mig2 repressor.BMC Cell Biol. 2012 Nov 6;13:31. doi: 10.1186/1471-2121-13-31. BMC Cell Biol. 2012. PMID: 23131016 Free PMC article.
-
Spatial regulation of the start repressor Whi5.Cell Cycle. 2009 Sep 15;8(18):3010-8. Epub 2009 Sep 25. Cell Cycle. 2009. PMID: 19713766
-
Nuclear localization signals for four distinct karyopherin-β nuclear import systems.Biochem J. 2015 Jun 15;468(3):353-62. doi: 10.1042/BJ20150368. Biochem J. 2015. PMID: 26173234 Review.
-
Nuclear import by karyopherin-βs: recognition and inhibition.Biochim Biophys Acta. 2011 Sep;1813(9):1593-606. doi: 10.1016/j.bbamcr.2010.10.014. Epub 2010 Oct 26. Biochim Biophys Acta. 2011. PMID: 21029754 Free PMC article. Review.
Cited by
-
Molecular basis of the functional distinction between Cln1 and Cln2 cyclins.Cell Cycle. 2012 Aug 15;11(16):3117-31. doi: 10.4161/cc.21505. Epub 2012 Aug 14. Cell Cycle. 2012. PMID: 22889732 Free PMC article.
-
Exportin Crm1 is important for Swi6 nuclear shuttling and MBF transcription activation in Saccharomyces cerevisiae.BMC Mol Cell Biol. 2022 Feb 21;23(1):10. doi: 10.1186/s12860-022-00409-6. BMC Mol Cell Biol. 2022. PMID: 35189816 Free PMC article.
-
Nuclear transporters in a multinucleated organism: functional and localization analyses in Aspergillus nidulans.Mol Biol Cell. 2011 Oct;22(20):3874-86. doi: 10.1091/mbc.E11-03-0262. Epub 2011 Aug 31. Mol Biol Cell. 2011. PMID: 21880896 Free PMC article.
-
Time-course proteome analysis reveals the dynamic response of Cryptococcus gattii cells to fluconazole.PLoS One. 2012;7(8):e42835. doi: 10.1371/journal.pone.0042835. Epub 2012 Aug 6. PLoS One. 2012. PMID: 22880118 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

