Nutlin-3, the small-molecule inhibitor of MDM2, promotes senescence and radiosensitises laryngeal carcinoma cells harbouring wild-type p53
- PMID: 20588277
- PMCID: PMC2906734
- DOI: 10.1038/sj.bjc.6605739
Nutlin-3, the small-molecule inhibitor of MDM2, promotes senescence and radiosensitises laryngeal carcinoma cells harbouring wild-type p53
Abstract
Background: Primary radiotherapy (RT) is a mainstay of treatment for laryngeal squamous cell carcinoma (LSCC). Although the cure rates for early (T1) vocal cord tumours are high, RT proves ineffective in up to a third of T3 carcinomas. Moreover, RT is associated with debilitating early- and late-treatment-related toxicity, thus finding means to de-escalate therapy, while retaining/augmenting therapeutic effectiveness, is highly desirable. p53 is a key mediator of radiation responses; we therefore investigated whether Nutlin-3, a small-molecule inhibitor of MDM2 (mouse double minute 2; an essential negative regulator of p53), might radiosensitise LSCC cells.
Methods: We performed clonogenic assays to measure radiosensitivity in a panel of LSCC cell lines (for which we determined p53 mutational status) in the presence and absence of Nutlin-3.
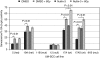
Results: LSCC cells harbouring wild-type p53 were significantly radiosensitised by Nutlin-3 (P<0.0001; log-rank scale), and displayed increased cell cycle arrest and significantly increased senescence (P<0.001) in the absence of increased apoptosis; thus, our data suggest that senescence may mediate this increased radiosensitivity.
Conclusion: This is the first study showing Nutlin-3 as an effective radiosensitiser in LSCC cells that retain wild-type p53. The clinical application of Nutlin-3 might improve local recurrence rates or allow treatment de-escalation in these patients.
Figures






Similar articles
-
Nutlin-3, an Antagonist of MDM2, Enhances the Radiosensitivity of Esophageal Squamous Cancer with Wild-Type p53.Pathol Oncol Res. 2018 Jan;24(1):75-81. doi: 10.1007/s12253-017-0215-5. Epub 2017 Mar 24. Pathol Oncol Res. 2018. PMID: 28341911
-
Radiosensitization of lung cancer by nutlin, an inhibitor of murine double minute 2.Mol Cancer Ther. 2006 Feb;5(2):411-7. doi: 10.1158/1535-7163.MCT-05-0356. Mol Cancer Ther. 2006. PMID: 16505116
-
Activation of p53 with Nutlin-3a radiosensitizes lung cancer cells via enhancing radiation-induced premature senescence.Lung Cancer. 2013 Aug;81(2):167-73. doi: 10.1016/j.lungcan.2013.04.017. Epub 2013 May 16. Lung Cancer. 2013. PMID: 23683497 Free PMC article.
-
Recent advances in the therapeutic perspectives of Nutlin-3.Curr Pharm Des. 2011;17(6):569-77. doi: 10.2174/138161211795222586. Curr Pharm Des. 2011. PMID: 21391907 Review.
-
Nutlins and ionizing radiation in cancer therapy.Curr Pharm Des. 2010;16(12):1427-42. doi: 10.2174/138161210791033932. Curr Pharm Des. 2010. PMID: 20166982 Review.
Cited by
-
Pre-clinical efficacy and synergistic potential of the MDM2-p53 antagonists, Nutlin-3 and RG7388, as single agents and in combined treatment with cisplatin in ovarian cancer.Oncotarget. 2016 Jun 28;7(26):40115-40134. doi: 10.18632/oncotarget.9499. Oncotarget. 2016. PMID: 27223080 Free PMC article.
-
Restoring p53 Function in Head and Neck Squamous Cell Carcinoma to Improve Treatments.Front Oncol. 2022 Jan 6;11:799993. doi: 10.3389/fonc.2021.799993. eCollection 2021. Front Oncol. 2022. PMID: 35071005 Free PMC article. Review.
-
BH3 mimetics selectively eliminate chemotherapy-induced senescent cells and improve response in TP53 wild-type breast cancer.Cell Death Differ. 2020 Nov;27(11):3097-3116. doi: 10.1038/s41418-020-0564-6. Epub 2020 May 26. Cell Death Differ. 2020. PMID: 32457483 Free PMC article.
-
Pinning Control for the p53-Mdm2 Network Dynamics Regulated by p14ARF.Front Physiol. 2020 Aug 28;11:976. doi: 10.3389/fphys.2020.00976. eCollection 2020. Front Physiol. 2020. PMID: 32982771 Free PMC article.
-
The Benefit of Reactivating p53 under MAPK Inhibition on the Efficacy of Radiotherapy in Melanoma.Cancers (Basel). 2019 Aug 1;11(8):1093. doi: 10.3390/cancers11081093. Cancers (Basel). 2019. PMID: 31374895 Free PMC article.
References
-
- Bernhard EJ, Maity A, Muschel RJ, McKenna WG (1995) Effects of ionizing radiation on cell cycle progression. A review. Radiat Environ Biophys 34: 79–83 - PubMed
-
- Berrino FGG (1998) Variation in survival of patients with head and neck cancer in Europe by the site of origin of the tumours. EUROCARE Working Group. Eur J Cancer 34: 2154–2161 - PubMed
-
- Bode AM, Dong Z (2004) Post-translational modification of p53 in tumorigenesis. Nat Rev Cancer 4: 793–805 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

