Prophylactic treatment with a G glycoprotein monoclonal antibody reduces pulmonary inflammation in respiratory syncytial virus (RSV)-challenged naive and formalin-inactivated RSV-immunized BALB/c mice
- PMID: 20592094
- PMCID: PMC2937657
- DOI: 10.1128/JVI.00451-10
Prophylactic treatment with a G glycoprotein monoclonal antibody reduces pulmonary inflammation in respiratory syncytial virus (RSV)-challenged naive and formalin-inactivated RSV-immunized BALB/c mice
Abstract
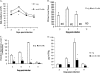
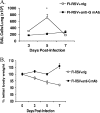
We examined whether prophylactically administered anti-respiratory syncytial virus (anti-RSV) G monoclonal antibody (MAb) would decrease the pulmonary inflammation associated with primary RSV infection and formalin-inactivated RSV (FI-RSV)-enhanced disease in mice. MAb 131-2G administration 1 day prior to primary infection reduced the pulmonary inflammatory response and the level of RSV replication. Further, intact or F(ab')(2) forms of MAb 131-2G administered 1 day prior to infection in FI-RSV-vaccinated mice reduced enhanced inflammation and disease. This study shows that an anti-RSV G protein MAb might provide prophylaxis against both primary infection and FI-RSV-associated enhanced disease. It is possible that antibodies with similar reactivities might prevent enhanced disease and improve the safety of nonlive virus vaccines.
Figures

Similar articles
-
Decrease in formalin-inactivated respiratory syncytial virus (FI-RSV) enhanced disease with RSV G glycoprotein peptide immunization in BALB/c mice.PLoS One. 2013 Dec 23;8(12):e83075. doi: 10.1371/journal.pone.0083075. eCollection 2013. PLoS One. 2013. PMID: 24376637 Free PMC article.
-
A respiratory syncytial virus (RSV) anti-G protein F(ab')2 monoclonal antibody suppresses mucous production and breathing effort in RSV rA2-line19F-infected BALB/c mice.J Virol. 2013 Oct;87(20):10955-67. doi: 10.1128/JVI.01164-13. Epub 2013 Jul 24. J Virol. 2013. PMID: 23885067 Free PMC article.
-
Prophylaxis with a respiratory syncytial virus (RSV) anti-G protein monoclonal antibody shifts the adaptive immune response to RSV rA2-line19F infection from Th2 to Th1 in BALB/c mice.J Virol. 2014 Sep;88(18):10569-83. doi: 10.1128/JVI.01503-14. Epub 2014 Jul 2. J Virol. 2014. PMID: 24990999 Free PMC article.
-
Prevention of Respiratory Syncytial Virus Infection: From Vaccine to Antibody.Microbiol Spectr. 2014 Aug;2(4):AID-0014-2014. doi: 10.1128/microbiolspec.AID-0014-2014. Microbiol Spectr. 2014. PMID: 26104207 Review.
-
New insights for development of a safe and protective RSV vaccine.Hum Vaccin. 2010 Jun;6(6):482-92. doi: 10.4161/hv.6.6.11562. Epub 2010 Jun 1. Hum Vaccin. 2010. PMID: 20671419 Free PMC article. Review.
Cited by
-
Anti-G protein antibodies targeting the RSV G protein CX3C chemokine region improve the interferon response.Ther Adv Infect Dis. 2023 Mar 14;10:20499361231161157. doi: 10.1177/20499361231161157. eCollection 2023 Jan-Dec. Ther Adv Infect Dis. 2023. PMID: 36938145 Free PMC article.
-
Comparisons of Antibody Populations in Different Pre-Fusion F VLP-Immunized Cotton Rat Dams and Their Offspring.Vaccines (Basel). 2020 Mar 18;8(1):133. doi: 10.3390/vaccines8010133. Vaccines (Basel). 2020. PMID: 32197348 Free PMC article.
-
Immune Prophylaxis Targeting the Respiratory Syncytial Virus (RSV) G Protein.Viruses. 2023 Apr 27;15(5):1067. doi: 10.3390/v15051067. Viruses. 2023. PMID: 37243153 Free PMC article.
-
Intranasal Vaccination with a Respiratory-Syncytial-Virus-Based Virus-like Particle Displaying the G Protein Conserved Region Induces Severe Weight Loss and Pathology upon Challenge with Wildtype Respiratory Syncytial Virus.Viruses. 2024 May 24;16(6):843. doi: 10.3390/v16060843. Viruses. 2024. PMID: 38932136 Free PMC article.
-
The respiratory syncytial virus G protein conserved domain induces a persistent and protective antibody response in rodents.PLoS One. 2012;7(3):e34331. doi: 10.1371/journal.pone.0034331. Epub 2012 Mar 29. PLoS One. 2012. PMID: 22479601 Free PMC article.
References
-
- Castilow, E. M., M. R. Olson, and S. M. Varga. 2007. Understanding respiratory syncytial virus (RSV) vaccine-enhanced disease. Immunol. Res. 39:225-239. - PubMed
-
- Chin, J., R. L. Magoffin, L. A. Shearer, J. H. Schieble, and E. H. Lennette. 1969. Field evaluation of a respiratory syncytial virus vaccine and a trivalent parainfluenza virus vaccine in a pediatric population. Am. J. Epidemiol. 89:449-463. - PubMed
-
- Council on Children with Disabilities. 2009. Policy statements—Supplemental Security Income (SSI) for children and youth with disabilities. Pediatrics 124:1702-1708. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

