Mast cells and Th17 cells contribute to the lymphoma-associated pro-inflammatory microenvironment of angioimmunoblastic T-cell lymphoma
- PMID: 20595635
- PMCID: PMC2913370
- DOI: 10.2353/ajpath.2010.091286
Mast cells and Th17 cells contribute to the lymphoma-associated pro-inflammatory microenvironment of angioimmunoblastic T-cell lymphoma
Abstract
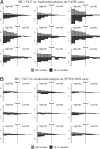
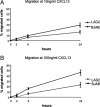
Reports focusing on the immunological microenvironment of peripheral T-cell lymphomas (PTCL) are rare. Here we studied the reciprocal contribution of regulatory (Treg) and interleukin-17-producing (Th17) T-cells to the composition of the lymphoma-associated microenvironment of angioimmunoblastic T-cell lymphoma (AITL) and PTCL not otherwise specified on tissue microarrays from 30 PTCLs not otherwise specified and 37 AITLs. We found that Th17 but not Treg cells were differently represented in the two lymphomas and correlated with the amount of mast cells (MCs) and granulocytes, which preferentially occurred in the cellular milieu of AITL cases. We observed that MCs directly synthesized interleukin-6 and thus contribute to the establishment of a pro-inflammatory, Th17 permissive environment in AITL. We further hypothesized that the AITL clone itself could be responsible for the preferential accumulation of MCs at sites of infiltration through the synthesis of CXCL-13 and its interaction with the CXCR3 and CXCR5 receptors expressed on MCs. Consistent with this hypothesis, we observed MCs efficiently migrating in response to CXCL-13. On these bases, we conclude that MCs have a role in molding the immunological microenvironment of AITL toward the maintenance of pro-inflammatory conditions prone to Th17 generation and autoimmunity.
Figures






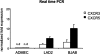
Similar articles
-
CD30 Expression by B and T Cells: A Frequent Finding in Angioimmunoblastic T-Cell Lymphoma and Peripheral T-Cell Lymphoma-Not Otherwise Specified.Am J Surg Pathol. 2016 Mar;40(3):378-85. doi: 10.1097/PAS.0000000000000571. Am J Surg Pathol. 2016. PMID: 26574847
-
High-dimensional and single-cell transcriptome analysis of the tumor microenvironment in angioimmunoblastic T cell lymphoma (AITL).Leukemia. 2022 Jan;36(1):165-176. doi: 10.1038/s41375-021-01321-2. Epub 2021 Jul 6. Leukemia. 2022. PMID: 34230608
-
Regulatory T-cell depletion in angioimmunoblastic T-cell lymphoma.Am J Pathol. 2010 Aug;177(2):570-4. doi: 10.2353/ajpath.2010.100150. Epub 2010 Jun 21. Am J Pathol. 2010. PMID: 20566750 Free PMC article.
-
Advances in understanding of angioimmunoblastic T-cell lymphoma.Leukemia. 2020 Oct;34(10):2592-2606. doi: 10.1038/s41375-020-0990-y. Epub 2020 Jul 23. Leukemia. 2020. PMID: 32704161 Free PMC article. Review.
-
Angioimmunoblastic T-cell lymphoma: the many-faced lymphoma.Blood. 2017 Mar 2;129(9):1095-1102. doi: 10.1182/blood-2016-09-692541. Epub 2017 Jan 23. Blood. 2017. PMID: 28115369 Review.
Cited by
-
Transcriptome sequencing reveals a profile that corresponds to genomic variants in Waldenström macroglobulinemia.Blood. 2016 Aug 11;128(6):827-38. doi: 10.1182/blood-2016-03-708263. Epub 2016 Jun 14. Blood. 2016. PMID: 27301862 Free PMC article.
-
Steering Mast Cells or Their Mediators as a Prospective Novel Therapeutic Approach for the Treatment of Hematological Malignancies.Front Oncol. 2021 Sep 24;11:731323. doi: 10.3389/fonc.2021.731323. eCollection 2021. Front Oncol. 2021. PMID: 34631562 Free PMC article. Review.
-
Role of cytokine in malignant T-cell metabolism and subsequent alternation in T-cell tumor microenvironment.Front Oncol. 2023 Sep 7;13:1235711. doi: 10.3389/fonc.2023.1235711. eCollection 2023. Front Oncol. 2023. PMID: 37746258 Free PMC article. Review.
-
Molecular genetics of peripheral T-cell lymphomas.Int J Hematol. 2014 Mar;99(3):219-26. doi: 10.1007/s12185-014-1522-1. Epub 2014 Jan 31. Int J Hematol. 2014. PMID: 24481943 Review.
-
Immune Microenvironment Features and Dynamics in Hodgkin Lymphoma.Cancers (Basel). 2021 Jul 20;13(14):3634. doi: 10.3390/cancers13143634. Cancers (Basel). 2021. PMID: 34298847 Free PMC article. Review.
References
-
- Vose J, Armitage J, Weisenburger D, International T-Cell Lymphoma Project International peripheral T-cell and natural killer/T-cell lymphoma study: pathology findings and clinical outcomes. J Clin Oncol. 2008;26:4124–4130. - PubMed
-
- de Leval L, Bisig B, Thielen C, Boniver J, Gaulard P. Molecular classification of T-cell lymphomas. Crit Rev Oncol Hematol. 2009;72:125–143. - PubMed
-
- de Leval L, Gaulard P. Pathobiology and molecular profiling of peripheral T-cell lymphomas. Hematology Am Soc Hematol Educ Program. 2008:272–279. - PubMed
-
- Carbone A, Gloghini A, Cabras A, Elia G. Differentiating germinal center-derived lymphomas through their cellular microenvironment. Am J Hematol. 2009;84:435–438. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

