Exercise capacity and idebenone intervention in children and adolescents with Friedreich ataxia
- PMID: 20599042
- PMCID: PMC2995920
- DOI: 10.1016/j.apmr.2010.04.007
Exercise capacity and idebenone intervention in children and adolescents with Friedreich ataxia
Abstract
Objective: To determine the exercise capacity of children and adolescents with Friedreich's Ataxia (FA) and to evaluate the effects of 6 months of idebenone treatment on exercise capacity.
Design: Exploratory endpoint in a randomized double-blind, placebo-controlled, phase II clinical trial designed to investigate the effects of idebenone on a biomarker of oxidative stress.
Setting: Exercise physiology laboratory in a single clinical research center.
Participants: Ambulatory subjects (N=48; age range, 9-17 y) with genetically confirmed FA.
Intervention: Idebenone administered orally 3 times a day for a total daily dose of approximately 5, 15, and 45 mg/kg or matching placebo for 6 months.
Main outcome measures: Peak oxygen consumption per unit time (peak VO(2)) and peak work rate (WR) were measured during incremental exercise testing at baseline and after treatment. Echocardiography and neurologic assessments were also completed before and after treatment.
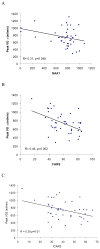
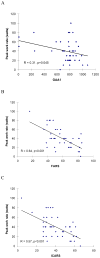
Results: Baseline mean peak VO(2) +/- SD was 746+/-246 mL/min (16.2+/-5.8 mL/kg/min), and WR was 40+/-23 W for all subjects. Peak VO(2) and WR were correlated with short guanine-adenine-adenine allele length and neurologic function. Relative left ventricular wall thickness was increased but left ventricular ejection fraction was normal in most subjects; there was no relationship between any exercise and echocardiographic measures. There were no significant changes in mean peak VO(2) or WR after idebenone treatment at any dose level relative to placebo.
Conclusions: Exercise capacity in children and adolescents with FA was significantly impaired. The basis for the impairment appears to be multifactorial and correlated to the degree of neurologic impairment. Although idebenone has previously been shown potentially to improve features of FA, idebenone treatment did not increase exercise capacity relative to placebo.
Trial registration: ClinicalTrials.gov NCT00229632.
Copyright 2010 American Congress of Rehabilitation Medicine. Published by Elsevier Inc. All rights reserved.
Figures

References
-
- Mateo I, Llorca J, Volpini V, Corral J, Berciano J, Combarros O. Expanded GAA repeats and clinical variation in Friedreich’s ataxia. Acta Neurol Scand. 2004;109(1):75–8. - PubMed
-
- Campuzano V, Montermini L, Molto MD, et al. Friedreich’s ataxia: autosomal recessive disease caused by an intronic GAA triplet repeat expansion. Science. 1996;271(5254):1423–7. - PubMed
-
- Durr A, Cossee M, Agid Y, et al. Clinical and genetic abnormalities in patients with Friedreich’s ataxia. N Engl J Med. 1996;335(16):1169–75. - PubMed
-
- Rotig A, de Lonlay P, Chretien D, et al. Aconitase and mitochondrial iron-sulphur protein deficiency in Friedreich ataxia. Nat Genet. 1997;17(2):215–7. - PubMed
-
- Schulz JB, Dehmer T, Schols L, et al. Oxidative stress in patients with Friedreich ataxia. Neurology. 2000;55(11):1719–21. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

