Dexamethasone transcriptionally increases the expression of the pregnane X receptor and synergistically enhances pyrethroid esfenvalerate in the induction of cytochrome P450 3A23
- PMID: 20599767
- PMCID: PMC2933965
- DOI: 10.1016/j.bcp.2010.06.043
Dexamethasone transcriptionally increases the expression of the pregnane X receptor and synergistically enhances pyrethroid esfenvalerate in the induction of cytochrome P450 3A23
Abstract
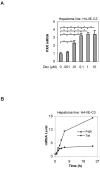
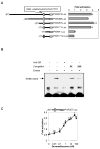
The pregnane X receptor (PXR) is recognized as a key regulator for the induction of a large number of genes in drug metabolism and transport. The transactivation of PXR is enhanced by the glucocorticoid dexamethasone and the enhancement is linked to the induction of PXR in humans and rats. The present study was undertaken to determine the mechanism for the induction and ascertain the synergistic effect on the expression of CYP3A23, a rat PXR target. In primary hepatocytes, significant induction of PXR was detected as early as 2h after the treatment and the maximal induction occurred at 1 microM dexamethasone. Similar induction kinetics was observed in the hepatoma line H4-II-E-C3. The induction was abolished by actinomycin D and dexamethasone efficaciously stimulated the rat PXR promoter. In addition, dexamethasone synergized esfenvalerate (an insecticide and a PXR activator) in inducing CYP3A23 and stimulating the CYP3A23 promoter. The full promoter of CYP3A23 (-1445/+74) was activated in a similar pattern as the changes in PXR mRNA in response to dexamethasone, esfenvalerate and co-treatment. In contrast, different responding patterns were detected on the stimulation of the CYP3A23 proximal promoter. Synergistic stimulation was also observed on the CYP3A4-DP-Luc reporter, the human counterpart of CYP3A23. These findings establish that transactivation is responsible for the induction of rat PXR and the induction presents potential interactions with insecticides in a species-conserved manner. The different responding patterns among CYP3A23 reporters point to an involvement of multiple transcriptional events in the regulation of CYP3A23 expression by dexamethasone, esfenvalerate and both.
Copyright 2010 Elsevier Inc. All rights reserved.
Figures




Similar articles
-
Two-stage glucocorticoid induction of CYP3A23 through both the glucocorticoid and pregnane X receptors.Mol Pharmacol. 2000 Jul;58(1):48-57. doi: 10.1124/mol.58.1.48. Mol Pharmacol. 2000. PMID: 10860926
-
Clofibrate and perfluorodecanoate both upregulate the expression of the pregnane X receptor but oppositely affect its ligand-dependent induction on cytochrome P450 3A23.Biochem Pharmacol. 2005 May 1;69(9):1363-71. doi: 10.1016/j.bcp.2005.02.011. Biochem Pharmacol. 2005. PMID: 15826607
-
Pregnane X receptor-dependent and -independent effects of 2-acetylaminofluorene on cytochrome P450 3A23 expression and liver cell proliferation.Biochem Biophys Res Commun. 2003 Jan 10;300(2):278-84. doi: 10.1016/s0006-291x(02)02847-4. Biochem Biophys Res Commun. 2003. PMID: 12504080
-
Simultaneous substitution of phenylalanine-305 and aspartate-318 of rat pregnane X receptor with the corresponding human residues abolishes the ability to transactivate the CYP3A23 promoter.J Pharmacol Exp Ther. 2005 Feb;312(2):571-82. doi: 10.1124/jpet.104.074971. Epub 2004 Sep 14. J Pharmacol Exp Ther. 2005. PMID: 15367577
-
Pregnane X receptor: molecular basis for species differences in CYP3A induction by xenobiotics.Chem Biol Interact. 2001 May 16;134(3):283-9. doi: 10.1016/s0009-2797(01)00163-6. Chem Biol Interact. 2001. PMID: 11336976 Review.
Cited by
-
Pregnane X receptor and drug-induced liver injury.Expert Opin Drug Metab Toxicol. 2014 Nov;10(11):1521-32. doi: 10.1517/17425255.2014.963555. Epub 2014 Sep 25. Expert Opin Drug Metab Toxicol. 2014. PMID: 25252616 Free PMC article. Review.
-
The immune response is a critical regulator of zebrafish retinal pigment epithelium regeneration.Proc Natl Acad Sci U S A. 2021 May 25;118(21):e2017198118. doi: 10.1073/pnas.2017198118. Proc Natl Acad Sci U S A. 2021. PMID: 34006636 Free PMC article.
-
Breast cancer-resistance protein (BCRP1) in the fetal mouse brain: development and glucocorticoid regulation.Biol Reprod. 2011 Apr;84(4):783-9. doi: 10.1095/biolreprod.110.088468. Epub 2010 Dec 15. Biol Reprod. 2011. PMID: 21159928 Free PMC article.
-
Glucocorticoid exposure induces preeclampsia via dampening 1,25-dihydroxyvitamin D3.Hypertens Res. 2018 Feb;41(2):104-111. doi: 10.1038/hr.2017.98. Epub 2017 Nov 2. Hypertens Res. 2018. PMID: 29093562
-
Retinal ganglion cell survival after severe optic nerve injury is modulated by crosstalk between Jak/Stat signaling and innate immune responses in the zebrafish retina.Development. 2022 Apr 15;149(8):dev199694. doi: 10.1242/dev.199694. Epub 2021 Oct 26. Development. 2022. PMID: 34528064 Free PMC article.
References
-
- Zhou SF. Drugs behave as substrates, inhibitors and inducers of human cytochrome P450 3A4. Curr Drug Metab. 2008;9:310–322. - PubMed
-
- Goodwin B, Redinbo MR, Kliewer SA. Regulation of cyp3a gene transcription by the pregnane x receptor. Annu Rev Pharmacol Toxicol. 2002;42:1–23. - PubMed
-
- Watkins RE, Wisely GB, Moore LB, Collins JL, Lambert MH, Williams SP, Willson TM, Kliewer SA, Redinbo MR. The human nuclear xenobiotic receptor PXR: structural determinants of directed promiscuity. Science. 2001;292:2329–2333. - PubMed
-
- Watkins RE, Maglich JM, Moore LB, Wisely GB, Noble SM, Davis-Searles PR, Lambert MH, Kliewer SA, Redinbo MR. 2.1 A crystal structure of human PXR in complex with the St. John’s wort compound hyperforin. Biochemistry. 2003;42:1430–1438. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

