Genome-wide analysis of germ cell proliferation in C.elegans identifies VRK-1 as a key regulator of CEP-1/p53
- PMID: 20599896
- PMCID: PMC3375680
- DOI: 10.1016/j.ydbio.2010.06.022
Genome-wide analysis of germ cell proliferation in C.elegans identifies VRK-1 as a key regulator of CEP-1/p53
Abstract
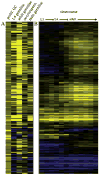
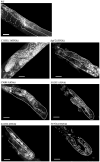
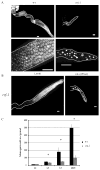
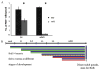
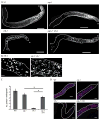
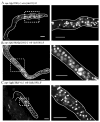
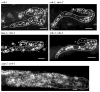
Proliferating germ cells in Caenorhabditiselegans provide a useful model system for deciphering fundamental mechanisms underlying the balance between proliferation and differentiation. Using gene expression profiling, we identified approximately 200 genes upregulated in the proliferating germ cells of C. elegans. Functional characterization using RNA-mediated interference demonstrated that over forty of these factors are required for normal germline proliferation and development. Detailed analysis of two of these factors defined an important regulatory relationship controlling germ cell proliferation. We established that the kinase VRK-1 is required for normal germ cell proliferation, and that it acts in part to regulate CEP-1(p53) activity. Loss of cep-1 significantly rescued the proliferation defects of vrk-1 mutants. We suggest that VRK-1 prevents CEP-1 from triggering an inappropriate cell cycle arrest, thereby promoting germ cell proliferation. This finding reveals a previously unsuspected mechanism for negative regulation of p53 activity in germ cells to control proliferation.
Copyright 2010 Elsevier Inc. All rights reserved.
Figures





References
-
- Aggarwal BD, Calvi BR. Chromatin regulates origin activity in Drosophila follicle cells. Nature. 2004;430:372–376. - PubMed
-
- Aladjem MI, Spike BT, Rodewald LW, Hope TJ, Klemm M, Jaenisch R, Wahl GM. ES cells do not activate p53-dependent stress responses and undergo p53-independent apoptosis in response to DNA damage. Curr Biol. 1998;8:145–155. - PubMed
-
- Berry LW, Westlund B, Schedl T. Germ-line tumor formation caused by activation of glp-1, a Caenorhabditis elegans member of the Notch family of receptors. Development. 1997;124:925–936. - PubMed
-
- Boehme KA, Blattner C. Regulation of p53–insights into a complex process. Crit Rev Biochem Mol Biol. 2009;44:367–392. - PubMed
-
- Bosl GJ, Motzer RJ. Testicular germ-cell cancer. N Engl J Med. 1997;337:242–253. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

