An analytical model for genetic hitchhiking in the evolution of antimalarial drug resistance
- PMID: 20600206
- PMCID: PMC2916054
- DOI: 10.1016/j.tpb.2010.06.005
An analytical model for genetic hitchhiking in the evolution of antimalarial drug resistance
Abstract
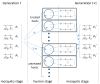
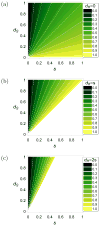
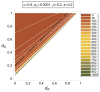
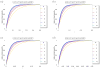
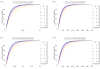
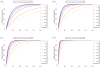
We analytically study a deterministic model for the spread of drug resistance among human malaria parasites. The model incorporates all major characteristics of the complex malaria transmission cycle and accounts for the fact that only a fraction alpha of infected hosts receive drug treatment. Furthermore, the model incorporates that hosts can be co-infected. The number m of parasites co-infecting a host is either a constant or, more generally, follows a given frequency distribution. Although the model is formulated in a multilocus setup, for our results we assume that drug resistance is caused by a single locus with two alleles - a sensitive one and a resistant one. We assume that the resistant allele has a selective advantage only in treated hosts and pays metabolic costs, which causes this allele to be deleterious in untreated hosts. We provide necessary and sufficient conditions for the fixation of the resistant allele. Moreover, provided the resistant allele will sweep through the population, we derive a formula for the time until it reaches a given frequency and in particular for the time until quasi-fixation. Furthermore, we establish an analytical solution for allele frequency changes at a linked neutral biallelic locus due to the rapid increase in frequency of the resistant allele. Our solution describes a local reduction in heterozygosity among parasite chromosomes around the resistant allele, the effect commonly referred to as the hitchhiking effect, as a function of alpha and m. The result therefore allows the investigation of selective sweep patterns under specific demographic settings. We find that the hitchhiking effect is similar but different from the standard model of genetic hitchhiking that assumes random mating and homogeneous selection. In particular, the process of recombination and selection cannot be decoupled. We further explain why standard hitchhiking theory cannot be applied to drug resistance in malaria. Furthermore, we will show that a genome-wide reduction in relative heterozygosity can occur provided a fraction of hosts is infected by a single parasite haplotype. Finally, we show how to incorporate host heterogeneity, and generalize our results to this biologically more realistic case.
Copyright 2010 Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Approximations for the hitchhiking effect caused by the evolution of antimalarial-drug resistance.J Math Biol. 2011 Jun;62(6):789-832. doi: 10.1007/s00285-010-0353-9. Epub 2010 Jul 11. J Math Biol. 2011. PMID: 20623287 Free PMC article.
-
The Rare, the Best: Spread of Antimalarial-Resistant Plasmodium falciparum Parasites by Anopheles Mosquito Vectors.Microbiol Spectr. 2021 Oct 31;9(2):e0085221. doi: 10.1128/Spectrum.00852-21. Epub 2021 Oct 20. Microbiol Spectr. 2021. PMID: 34668767 Free PMC article.
-
Plasmodium falciparum: differential selection of drug resistance alleles in contiguous urban and peri-urban areas of Brazzaville, Republic of Congo.PLoS One. 2011;6(8):e23430. doi: 10.1371/journal.pone.0023430. Epub 2011 Aug 15. PLoS One. 2011. PMID: 21858115 Free PMC article.
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
-
The selection landscape of malaria parasites.Science. 2010 May 14;328(5980):866-71. doi: 10.1126/science.1185410. Science. 2010. PMID: 20466925 Review.
Cited by
-
Fitness components and natural selection: why are there different patterns on the emergence of drug resistance in Plasmodium falciparum and Plasmodium vivax?Malar J. 2013 Jan 11;12:15. doi: 10.1186/1475-2875-12-15. Malar J. 2013. PMID: 23305428 Free PMC article.
-
Evolutionary genetics of malaria.Front Genet. 2022 Nov 3;13:1030463. doi: 10.3389/fgene.2022.1030463. eCollection 2022. Front Genet. 2022. PMID: 36406132 Free PMC article.
-
A population genetic model for the initial spread of partially resistant malaria parasites under anti-malarial combination therapy and weak intrahost competition.PLoS One. 2014 Jul 9;9(7):e101601. doi: 10.1371/journal.pone.0101601. eCollection 2014. PLoS One. 2014. PMID: 25007207 Free PMC article.
-
Genetic hitchhiking under heterogeneous spatial selection pressures.PLoS One. 2013 Apr 24;8(4):e61742. doi: 10.1371/journal.pone.0061742. Print 2013. PLoS One. 2013. PMID: 23637897 Free PMC article.
-
Local population structure of Plasmodium: impact on malaria control and elimination.Malar J. 2012 Dec 11;11:412. doi: 10.1186/1475-2875-11-412. Malar J. 2012. PMID: 23232077 Free PMC article.
References
-
- Anderson TJC, Haubold B, Williams JT, Estrada-Francosection JG, Richardson L, Mollinedo R, Bockarie M, Mokili J, Mharakurwa S, French N, Whitworth J, Velez ID, Brockman AH, Nosten F, Ferreira MU, Day KP. Microsatellite Markers Reveal a Spectrum of Population Structures in the Malaria Parasite Plasmodium falciparum. Mol Biol Evol. 2000. pp. 1467–1482. URL http://mbe.oxfordjournals.org/cgi/content/abstract/17/10/1467. - PubMed
-
- Arnot D. Clone multiplicity of plasmodium falciparum infections in individuals exposed to variable levels of disease transmission. Transactions of the Royal Society of Tropical Medicine and Hygiene. 1998. pp. 580–585. URL http://www.sciencedirect.com/science/article/B75GP-4C41SD8-7C/2/8c5ea9d3.... - PubMed
-
- Barton NH. Genetic hitchhiking. Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences. 2000. pp. 1553–1562. URL http://rstb.royalsocietypublishing.org/content/355/1403/1553.abstract. - PMC - PubMed
-
- Cornejo OE, Escalante AA. The origin and age of plasmodium vivax. Dec, 2006. URL http://linkinghub.elsevier.com/retrieve/pii/S147149220600256X. - PMC - PubMed
-
- Daily JP. Antimalarial Drug Therapy: The Role of Parasite Biology and Drug Resistance. J Clin Pharmacol. 2006. pp. 1487–1497. URL http://jcp.sagepub.com/cgi/content/abstract/46/12/1487. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

