Alzheimer disease identification using amyloid imaging and reserve variables: proof of concept
- PMID: 20603484
- PMCID: PMC2906402
- DOI: 10.1212/WNL.0b013e3181e620f4
Alzheimer disease identification using amyloid imaging and reserve variables: proof of concept
Abstract
Objective: Several factors may influence the relationship between Alzheimer disease (AD) lesions and the expression of dementia, including those related to brain and cognitive reserve. Other factors may confound the association between AD pathology and dementia. We tested whether factors thought to influence the association of AD pathology and dementia help to accurately identify dementia of the Alzheimer type (DAT) when considered together with amyloid imaging.
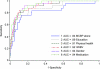
Methods: Participants with normal cognition (n = 180) and with DAT (n = 25), aged 50 years or older, took part in clinical, neurologic, and psychometric assessments. PET with the Pittsburgh compound B (PiB) tracer was used to measure brain amyloid, yielding a mean cortical binding potential (MCBP) reflecting PiB uptake. Logistic regression was used to generate receiver operating characteristic curves, and the areas under those curves (AUC), to compare the predictive accuracy of using MCBP alone vs MCBP together with other variables selected using a stepwise selection procedure to identify participants with DAT vs normal cognition.
Results: The AUC resulting from MCBP alone was 0.84 (95% confidence interval [CI] = 0.73-0.94; cross-validated AUC = 0.80, 95% CI = 0.68-0.92). The AUC for the predictive equation generated by a stepwise model including education, normalized whole brain volume, physical health rating, gender, and use of medications that may interfere with cognition was 0.94 (95% CI = 0.90-0.98; cross-validated AUC = 0.91, 95% CI = 0.85-0.96), an improvement (p = 0.025) over that yielded using MCBP alone.
Conclusion: Results suggest that factors reported to influence associations between AD pathology and dementia can improve the predictive accuracy of amyloid imaging for the identification of symptomatic AD.
Figures

References
-
- Katzman R, Terry R, DeTeresa R, et al. Clinical, pathological, and neurochemical changes in dementia: a subgroup with preserved mental status and numerous neocortical plaques. Ann Neurol 1988;23:138–144. - PubMed
-
- Crystal H, Dickson D, Fuld P, et al. Clinico-pathologic studies in dementia: nondemented subjects with pathologically confirmed Alzheimer's disease. Neurology 1988;38:1682–1687. - PubMed
-
- Price JL, Morris JC. Tangles and plaques in nondemented aging and “preclinical” Alzheimer's disease. Ann Neurol 1999;45:358–368. - PubMed
-
- Klunk WE, Engler H, Nordberg A, et al. Imaging brain amyloid in Alzheimer's disease with Pittsburgh Compound-B. Ann Neurol 2004;55:306–319. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P50 AG005681-22/AG/NIA NIH HHS/United States
- U01AG016976/AG/NIA NIH HHS/United States
- R01NS066905-01/NS/NINDS NIH HHS/United States
- P01AG03991/AG/NIA NIH HHS/United States
- R01 DC009095-03/DC/NIDCD NIH HHS/United States
- P01AG003991/AG/NIA NIH HHS/United States
- 5 R01 AG15928-02/AG/NIA NIH HHS/United States
- 1U24RR026057/RR/NCRR NIH HHS/United States
- 1UL1RR024992/RR/NCRR NIH HHS/United States
- 1RC1AG036045-01/AG/NIA NIH HHS/United States
- P50AG005681/AG/NIA NIH HHS/United States
- 1U24RR02573601/RR/NCRR NIH HHS/United States
- 1RC2MD004750-01/MD/NIMHD NIH HHS/United States
- 5U54EB00514905/EB/NIBIB NIH HHS/United States
- 1K12RR024994/RR/NCRR NIH HHS/United States
- P30 CA091842/CA/NCI NIH HHS/United States
- P30-NS048056/NS/NINDS NIH HHS/United States
- 1R01EB009352/EB/NIBIB NIH HHS/United States
- 1UL1RR024992-01/RR/NCRR NIH HHS/United States
- P01-AG03991/AG/NIA NIH HHS/United States
- P01AG026276/AG/NIA NIH HHS/United States
- T32 NS007205/NS/NINDS NIH HHS/United States
- 2P30NS048056-06/NS/NINDS NIH HHS/United States
- 1R01NS055963-01/NS/NINDS NIH HHS/United States
- R01AG16335/AG/NIA NIH HHS/United States
- 1U01AG032438-02/AG/NIA NIH HHS/United States
- U01AG024904/AG/NIA NIH HHS/United States
- U01 AG016976/AG/NIA NIH HHS/United States
- P01 AG003991/AG/NIA NIH HHS/United States
- P50 AG005681/AG/NIA NIH HHS/United States
- P30 NS048056-01/NS/NINDS NIH HHS/United States
- U01AG032438/AG/NIA NIH HHS/United States
- P01 AG026276/AG/NIA NIH HHS/United States
- UL1 RR024992/RR/NCRR NIH HHS/United States
- P30 NS048056/NS/NINDS NIH HHS/United States
- P50-AG05681/AG/NIA NIH HHS/United States
- KL2RR024994/RR/NCRR NIH HHS/United States
- 2P01 AG03991-26/AG/NIA NIH HHS/United States
- U54CA136398-02/CA/NCI NIH HHS/United States
- 5U01AG03243802/AG/NIA NIH HHS/United States
- UL1 TR000448/TR/NCATS NIH HHS/United States
- 5K12RR023249-03/RR/NCRR NIH HHS/United States
- P50 NS006833/NS/NINDS NIH HHS/United States
- U24AG026395/AG/NIA NIH HHS/United States
- P50NS006833/NS/NINDS NIH HHS/United States
- P50AG05681/AG/NIA NIH HHS/United States
- 3 R01 DK063202-03S1/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
