Doxycycline's effect on ocular angiogenesis: an in vivo analysis
- PMID: 20605212
- PMCID: PMC2934900
- DOI: 10.1016/j.ophtha.2010.01.037
Doxycycline's effect on ocular angiogenesis: an in vivo analysis
Abstract
Purpose: To determine the in vivo effect of doxycycline on choroidal angiogenesis and pterygium growth by using a choroidal neovascular (CNV) murine model, a directed in vivo angiogenesis assay (DIVAA) and a pterygium murine model.
Design: Experimental study.
Participants: Three murine models were investigated with 4 mice minimum per group and 22 maximum per group.
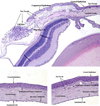
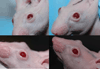
Methods: Mice received water with or without doxycycline. For the CNV, the neovascular lesion volume was determined in choroid-retinal pigment epithelial flat mounts using confocal microscopy 7 days after laser induction. For DIVAA, silicone capsules containing 10,000 human pterygium epithelial cells were implanted in the flanks of mice subcutaneously. After 11 days, neovascularization (NV) was quantified using spectrofluorometry after murine tail-vein injection of fluorescein isothiocyanate-labeled dextran. A pterygium epithelial cell model was developed by injecting 10,000 human pterygium epithelial cells in the nasal subconjunctival space in athymic nude mice. Doxycycline was started on day 6 at 50 mg/kg per day; corneal lesions that resulted from the injections were compared at days 6 and 15.
Main outcome measures: The Student t-test was used to evaluate the data for the CNV and DIVAA models and histologic preparations were used to evaluate pterygia lesions.
Results: There was significantly less NV and lesion volume with doxycycline taken in drinking water versus plain water. With doxycycline treatment, the laser-induced CNV showed a maximal 66% decrease in choroidal blood vessel volume (P< or =0.008) and the DIVAA showed a 30% reduction of blood vessel growth and migration (P<0.004). Histologic preparations demonstrated that pterygium cell lesions regressed when mice were administered doxycycline for 9 days.
Conclusions: Doxycycline significantly inhibited angiogenesis in 3 murine models. The most dramatic effect was found in the CNV model followed by the pterygia epithelial cell DIVAA model. The anterior segment pterygium model also showed regression histologically. This suggests that doxycycline may be successful as an adjunctive treatment for CNV and pterygia in humans; clinical trials would be necessary to determine if there is a benefit.
Copyright © 2010 American Academy of Ophthalmology. Published by Elsevier Inc. All rights reserved.
Figures








Comment in
-
Doxycycline's and ocular angiogenesis.Ophthalmology. 2011 Apr;118(4):789-90; author reply 790-1. doi: 10.1016/j.ophtha.2011.01.009. Ophthalmology. 2011. PMID: 21459227 No abstract available.
Similar articles
-
Doxycycline's and ocular angiogenesis.Ophthalmology. 2011 Apr;118(4):789-90; author reply 790-1. doi: 10.1016/j.ophtha.2011.01.009. Ophthalmology. 2011. PMID: 21459227 No abstract available.
-
Doxycycline-mediated inhibition of choroidal neovascularization.Invest Ophthalmol Vis Sci. 2009 Nov;50(11):5098-106. doi: 10.1167/iovs.08-3174. Epub 2009 Jun 10. Invest Ophthalmol Vis Sci. 2009. PMID: 19516001 Free PMC article.
-
Targeting immune privilege to prevent pathogenic neovascularization.Invest Ophthalmol Vis Sci. 2010 Jul;51(7):3560-6. doi: 10.1167/iovs.09-3890. Epub 2010 Feb 17. Invest Ophthalmol Vis Sci. 2010. PMID: 20164456 Free PMC article.
-
Suppression of choroidal neovascularization by vasohibin-1, a vascular endothelium-derived angiogenic inhibitor.Invest Ophthalmol Vis Sci. 2011 May 17;52(6):3272-80. doi: 10.1167/iovs.10-6295. Invest Ophthalmol Vis Sci. 2011. PMID: 21345982
-
Peroxisome proliferator-activated receptor-gamma ligands inhibit choroidal neovascularization.Invest Ophthalmol Vis Sci. 2000 Jul;41(8):2309-17. Invest Ophthalmol Vis Sci. 2000. PMID: 10892878
Cited by
-
Minocycline and Diacetyl Minocycline Eye Drops Reduce Ocular Neovascularization in Mice.Transl Vis Sci Technol. 2023 Dec 1;12(12):10. doi: 10.1167/tvst.12.12.10. Transl Vis Sci Technol. 2023. PMID: 38064336 Free PMC article.
-
Synthesis and antibacterial activity of doxycycline neoglycosides.J Nat Prod. 2013 Sep 27;76(9):1627-36. doi: 10.1021/np4003096. Epub 2013 Aug 29. J Nat Prod. 2013. PMID: 23987662 Free PMC article.
-
Ocular penetration of topically applied 1% tigecycline in a rabbit model.Int J Ophthalmol. 2017 May 18;10(5):679-683. doi: 10.18240/ijo.2017.05.03. eCollection 2017. Int J Ophthalmol. 2017. PMID: 28546920 Free PMC article.
-
Doxycycline prevents matrix remodeling and contraction by trichiasis-derived conjunctival fibroblasts.Invest Ophthalmol Vis Sci. 2013 Jul 12;54(7):4675-82. doi: 10.1167/iovs.13-11787. Invest Ophthalmol Vis Sci. 2013. PMID: 23766479 Free PMC article.
-
Key Clinical and Histopathological Features of a Pterygium-Like Induced Lesion in a Rabbit Model.Transl Vis Sci Technol. 2024 Oct 1;13(10):1. doi: 10.1167/tvst.13.10.1. Transl Vis Sci Technol. 2024. PMID: 39352714 Free PMC article.
References
-
- Campochiaro PA. Seeing the light: New insights into the molecular pathogenesis of retinal diseases. J Cell Physiol. 2007;213:348–354. - PubMed
-
- Steen B, Sejersen S, Berglin L, et al. Matrix metalloproteinases and metalloproteinase inhibitors in choroidal neovascular membranes. Invest Ophthalmol Vis Sci. 1998;39:2194–2200. - PubMed
-
- Dushku N, John MK, Schultz GS, Reid TW. Pterygia pathogenesis: corneal invasion by matrix metalloproteinase expressing altered limbal epithelial basal cells. Arch Ophthalmol. 2001;119:695–706. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

