The Arabidopsis plastid-signalling mutant gun1 (genomes uncoupled1) shows altered sensitivity to sucrose and abscisic acid and alterations in early seedling development
- PMID: 20605896
- PMCID: PMC2921207
- DOI: 10.1093/jxb/erq186
The Arabidopsis plastid-signalling mutant gun1 (genomes uncoupled1) shows altered sensitivity to sucrose and abscisic acid and alterations in early seedling development
Abstract
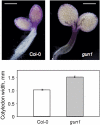
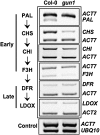
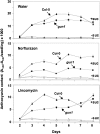
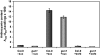
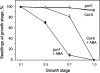
Developing seedlings of the Arabidopsis gun1 (genomes uncoupled1) mutant, which is defective in retrograde plastid-to-nucleus signalling, show several previously unrecognized mutant phenotypes. gun1 seedlings accumulated less anthocyanin than wild-type seedlings when grown in the presence of 2% (w/v) sucrose, due to lower amounts of transcripts of early anthocyanin biosynthesis genes in gun1. Norflurazon and lincomycin, which induce retrograde signalling, further decreased the anthocyanin content of sucrose-treated seedlings, and altered the temporal pattern of anthocyanin accumulation. Lincomycin treatment altered the spatial pattern of sucrose-induced anthocyanin accumulation, suggesting that plastids provide information for the regulation of anthocyanin biosynthesis in Arabidopsis seedlings. The temporal pattern of accumulation of LHCB1 transcripts differed between wild-type and gun1 seedlings, and gun1 seedlings were more sensitive to sucrose suppression of LHCB1 transcript accumulation than wild-type seedlings. Growth and development of gun1 seedlings was more sensitive to exogenous 2% sucrose than wild-type seedlings and, in the presence of lincomycin, cotyledon expansion was enhanced in gun1 seedlings compared to the wild type. gun1 seedlings were more sensitive than wild-type seedlings to the inhibition of seedling growth and development by abscisic acid. These observations clearly implicate GUN1 and plastid signalling in the regulation of seedling development and anthocyanin biosynthesis, and indicate a complex interplay between sucrose and plastid signalling pathways.
Figures




Similar articles
-
Timing the switch to phototrophic growth: a possible role of GUN1.Plant Signal Behav. 2011 Apr;6(4):578-82. doi: 10.4161/psb.6.4.14900. Plant Signal Behav. 2011. PMID: 21673514 Free PMC article.
-
A gain-of-function mutation of plastidic invertase alters nuclear gene expression with sucrose treatment partially via GENOMES UNCOUPLED1-mediated signaling.New Phytol. 2015 May;206(3):1013-1023. doi: 10.1111/nph.13309. Epub 2015 Jan 27. New Phytol. 2015. PMID: 25628228
-
The nuclear genes Lhcb and HEMA1 are differentially sensitive to plastid signals and suggest distinct roles for the GUN1 and GUN5 plastid-signalling pathways during de-etiolation.Plant J. 2004 Dec;40(5):672-85. doi: 10.1111/j.1365-313X.2004.02243.x. Plant J. 2004. PMID: 15546351
-
GUN control in retrograde signaling: How GENOMES UNCOUPLED proteins adjust nuclear gene expression to plastid biogenesis.Plant Cell. 2021 May 5;33(3):457-474. doi: 10.1093/plcell/koaa048. Plant Cell. 2021. PMID: 33955483 Free PMC article. Review.
-
The plastid transcription machinery and its coordination with the expression of nuclear genome: Plastid-Encoded Polymerase, Nuclear-Encoded Polymerase and the Genomes Uncoupled 1-mediated retrograde communication.Philos Trans R Soc Lond B Biol Sci. 2020 Jun 22;375(1801):20190399. doi: 10.1098/rstb.2019.0399. Epub 2020 May 4. Philos Trans R Soc Lond B Biol Sci. 2020. PMID: 32362266 Free PMC article. Review.
Cited by
-
genome uncoupled1 Mutants Are Hypersensitive to Norflurazon and Lincomycin.Plant Physiol. 2018 Nov;178(3):960-964. doi: 10.1104/pp.18.00772. Epub 2018 Aug 28. Plant Physiol. 2018. PMID: 30154176 Free PMC article.
-
Sucrose signaling in plants: a world yet to be explored.Plant Signal Behav. 2013 Mar;8(3):e23316. doi: 10.4161/psb.23316. Epub 2013 Jan 18. Plant Signal Behav. 2013. PMID: 23333971 Free PMC article. Review.
-
The roles of two transcription factors, ABI4 and CBFA, in ABA and plastid signalling and stress responses.Plant Mol Biol. 2013 Nov;83(4-5):445-58. doi: 10.1007/s11103-013-0102-8. Epub 2013 Jul 6. Plant Mol Biol. 2013. PMID: 23832569
-
Plastids are major regulators of light signaling in Arabidopsis.Plant Physiol. 2012 May;159(1):366-90. doi: 10.1104/pp.112.193599. Epub 2012 Mar 1. Plant Physiol. 2012. PMID: 22383539 Free PMC article.
-
ABI4 and its role in chloroplast retrograde communication.Front Plant Sci. 2013 Jan 10;3:304. doi: 10.3389/fpls.2012.00304. eCollection 2012. Front Plant Sci. 2013. PMID: 23335930 Free PMC article.
References
-
- Abdallah F, Salamini F, Leister D. A prediction of the size and evolutionary origin of the proteome of chloroplasts of Arabidopsis. Trends in Plant Sciences. 2000;5:141–142. - PubMed
-
- Acevedo-Hernandez GJ, Leon P, Herrera-Estrella LR. Sugar and ABA responsiveness of a minimal RBCS light-responsive unit is mediated by direct binding of ABI4. The Plant Journal. 2005;43:506–519. - PubMed
-
- Batschauer A, Mösinger E, Kreuz K, Dörr I, Apel K. The implication of a plastid-derived factor in the transcriptional control of nuclear genes encoding the light-harvesting chlorophyll a/b protein. European Journal of Biochemistry. 1986;154:625–634. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

