Role of progerin-induced telomere dysfunction in HGPS premature cellular senescence
- PMID: 20605919
- PMCID: PMC2908049
- DOI: 10.1242/jcs.067306
Role of progerin-induced telomere dysfunction in HGPS premature cellular senescence
Abstract
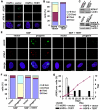
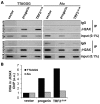
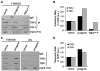
Hutchinson-Gilford Progeria Syndrome (HGPS) is a premature-aging syndrome caused by a dominant mutation in the gene encoding lamin A, which leads to an aberrantly spliced and processed protein termed progerin. Previous studies have shown that progerin induces early senescence associated with increased DNA-damage signaling and that telomerase extends HGPS cellular lifespan. We demonstrate that telomerase extends HGPS cellular lifespan by decreasing progerin-induced DNA-damage signaling and activation of p53 and Rb pathways that otherwise mediate the onset of premature senescence. We show further that progerin-induced DNA-damage signaling is localized to telomeres and is associated with telomere aggregates and chromosomal aberrations. Telomerase amelioration of DNA-damage signaling is relatively rapid, requires both its catalytic and DNA-binding functions, and correlates in time with the acquisition by HGPS cells of the ability to proliferate. All of these findings establish that HGPS premature cellular senescence results from progerin-induced telomere dysfunction.
Figures




References
-
- Amiel A., Fejgin M. D., Goldberg-Bittman L., Sharoni R., Hadary R., Kitay-Cohen Y. (2009). Telomere aggregates in hepatitis C patients. Cancer Invest. 27, 650-654 - PubMed
-
- Bolzan A. D., Bianchi M. S. (2006). Telomeres, interstitial telomeric repeat sequences, and chromosomal aberrations. Mutat. Res. 612, 189-214 - PubMed
-
- Callen E., Surralles J. (2004). Telomere dysfunction in genome instability syndromes. Mutat. Res. 567, 85-104 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

