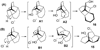
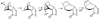Strained to the limit: when a cyclobutyl moiety becomes a thermodynamic sink in a protolytic ring-opening of photogenerated oxetanes
- PMID: 20608636
- PMCID: PMC2912957
- DOI: 10.1021/ol101297b
Strained to the limit: when a cyclobutyl moiety becomes a thermodynamic sink in a protolytic ring-opening of photogenerated oxetanes
Abstract
Strained polycyclic oxetanes generated photochemically from the Diels-Alder adducts of cyclic dienes and enones undergo deep skeletal rearrangements under protolytic ring-opening conditions offering expeditious access to chlorohydrins and other products of unique skeletal topology.
Figures
References
-
- Sauers RR, Kelly KW. J. Org. Chem. 1970;35:498–501.
- Sauers RR, Whittle JA. J. Org. Chem. 1969;34:3579–3582.
- Sauers RR, Schinski W, Mason MM. Tetrahedron Lett. 1969:79–82.
- Sauers RR, Kelly KW, Sickles BR. J. Org. Chem. 1972;37:537–543.
- Sauers RR. J. Org. Chem. 1974;39:1850–1853.
-
- Rawal VH, Dufour C. J. Am. Chem. Soc. 1994;116:2613–2614.
- Dvorak CA, Dufour C, Iwasa S, Rawal VH. J. Org. Chem. 1998;63:5302–5303.
-
- Valiulin RA, Kutateladze AG. Org. Lett. 2009;11:3886–3889. - PubMed
-
- Pérez-Ruiza R, Miranda MA, Alleb R, Meerholzb K, Griesbeck AG. Photochem. Photobiol. Sci. 2006;5:51–55. - PubMed
-
- Warrener RN, McCay IW, Paddon-Row MN. Aust. J. Chem. 1977;30:89–94.
- Coxon JM, O’Connell MJ, Steel PJ. Aust. J. Chem. 1986;39:1537–1557.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources