FIP1/RCP binding to Golgin-97 regulates retrograde transport from recycling endosomes to the trans-Golgi network
- PMID: 20610657
- PMCID: PMC2929997
- DOI: 10.1091/mbc.E10-04-0313
FIP1/RCP binding to Golgin-97 regulates retrograde transport from recycling endosomes to the trans-Golgi network
Abstract
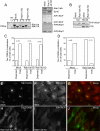
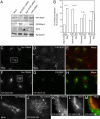
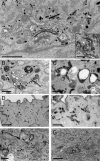
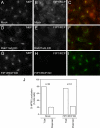
Many proteins are retrieved to the trans-Golgi Network (TGN) from the endosomal system through several retrograde transport pathways to maintain the composition and function of the TGN. However, the molecular mechanisms involved in these distinct retrograde pathways remain to be fully understood. Here we have used fluorescence and electron microscopy as well as various functional transport assays to show that Rab11a/b and its binding protein FIP1/RCP are both required for the retrograde delivery of TGN38 and Shiga toxin from early/recycling endosomes to the TGN, but not for the retrieval of mannose-6-phosphate receptor from late endosomes. Furthermore, by proteomic analysis we identified Golgin-97 as a FIP1/RCP-binding protein. The FIP1/RCP-binding domain maps to the C-terminus of Golgin-97, adjacent to its GRIP domain. Binding of FIP1/RCP to Golgin-97 does not affect Golgin-97 recruitment to the TGN, but appears to regulate the targeting of retrograde transport vesicles to the TGN. Thus, we propose that FIP1/RCP binding to Golgin-97 is required for tethering and fusion of recycling endosome-derived retrograde transport vesicles to the TGN.
Figures





Similar articles
-
The golgin GCC88 is required for efficient retrograde transport of cargo from the early endosomes to the trans-Golgi network.Mol Biol Cell. 2007 Dec;18(12):4979-91. doi: 10.1091/mbc.e07-06-0622. Epub 2007 Oct 3. Mol Biol Cell. 2007. PMID: 17914056 Free PMC article.
-
Identification of different itineraries and retromer components for endosome-to-Golgi transport of TGN38 and Shiga toxin.Eur J Cell Biol. 2010 May;89(5):379-93. doi: 10.1016/j.ejcb.2009.10.021. Epub 2010 Feb 6. Eur J Cell Biol. 2010. PMID: 20138391
-
Rab11 regulates the compartmentalization of early endosomes required for efficient transport from early endosomes to the trans-golgi network.J Cell Biol. 2000 Dec 11;151(6):1207-20. doi: 10.1083/jcb.151.6.1207. J Cell Biol. 2000. PMID: 11121436 Free PMC article.
-
Retrograde transport from endosomes to the trans-Golgi network.Nat Rev Mol Cell Biol. 2006 Aug;7(8):568-79. doi: 10.1038/nrm1985. Nat Rev Mol Cell Biol. 2006. PMID: 16936697 Review.
-
The regulation of endosome-to-Golgi retrograde transport by tethers and scaffolds.Traffic. 2011 Aug;12(8):939-47. doi: 10.1111/j.1600-0854.2011.01185.x. Epub 2011 Apr 8. Traffic. 2011. PMID: 21477175 Review.
Cited by
-
Different localization of lysosomal-associated membrane protein 1 (LAMP1) in mammalian cultured cell lines.Histochem Cell Biol. 2020 Apr;153(4):199-213. doi: 10.1007/s00418-019-01842-z. Epub 2020 Jan 6. Histochem Cell Biol. 2020. PMID: 31907597
-
EHD1 and RUSC2 Control Basal Epidermal Growth Factor Receptor Cell Surface Expression and Recycling.Mol Cell Biol. 2020 Mar 16;40(7):e00434-19. doi: 10.1128/MCB.00434-19. Print 2020 Mar 16. Mol Cell Biol. 2020. PMID: 31932478 Free PMC article.
-
Transport Vesicle Tethering at the Trans Golgi Network: Coiled Coil Proteins in Action.Front Cell Dev Biol. 2016 Mar 15;4:18. doi: 10.3389/fcell.2016.00018. eCollection 2016. Front Cell Dev Biol. 2016. PMID: 27014693 Free PMC article. Review.
-
Downregulation of ARID1A, a component of the SWI/SNF chromatin remodeling complex, in breast cancer.J Cancer. 2017 Jan 1;8(1):1-8. doi: 10.7150/jca.16602. eCollection 2017. J Cancer. 2017. PMID: 28123592 Free PMC article.
-
Rac1-Rab11-FIP3 regulatory hub coordinates vesicle traffic with actin remodeling and T-cell activation.EMBO J. 2016 Jun 1;35(11):1160-74. doi: 10.15252/embj.201593274. Epub 2016 May 6. EMBO J. 2016. PMID: 27154205 Free PMC article.
References
-
- Belenkaya T. Y., Wu Y., Tang X., Zhou B., Cheng L., Sharma Y. V., Yan D., Selva E. M., Lin X. The retromer complex influences Wnt secretion by recycling wntless from endosomes to the trans-Golgi network. Dev. Cell. 2008;14:120–131. - PubMed
-
- Bonifacino J. S., Rojas R. Retrograde transport from endosomes to the trans-Golgi network. Nat. Rev. Mol. Cell Biol. 2006;7:568–579. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

