T cell responses are required for protection from clinical disease and for virus clearance in severe acute respiratory syndrome coronavirus-infected mice
- PMID: 20610717
- PMCID: PMC2937604
- DOI: 10.1128/JVI.01049-10
T cell responses are required for protection from clinical disease and for virus clearance in severe acute respiratory syndrome coronavirus-infected mice
Abstract
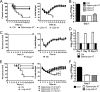
A dysregulated innate immune response and exuberant cytokine/chemokine expression are believed to be critical factors in the pathogenesis of severe acute respiratory syndrome (SARS), caused by a coronavirus (SARS-CoV). However, we recently showed that inefficient immune activation and a poor virus-specific T cell response underlie severe disease in SARS-CoV-infected mice. Here, we extend these results to show that virus-specific T cells, in the absence of activation of the innate immune response, were sufficient to significantly enhance survival and diminish clinical disease. We demonstrated that T cells are responsible for virus clearance, as intravenous adoptive transfer of SARS-CoV-immune splenocytes or in vitro-generated T cells to SCID or BALB/c mice enhanced survival and reduced virus titers in the lung. Enhancement of the number of virus-specific CD8 T cells by immunization with SARS-CoV peptide-pulsed dendritic cells also resulted in a robust T cell response, earlier virus clearance, and increased survival. These studies are the first to show that T cells play a crucial role in SARS-CoV clearance and that a suboptimal T cell response contributes to the pathological changes observed in SARS. They also provide a new approach to SARS vaccine design.
Figures

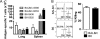


References
-
- Badovinac, V. P., K. A. Messingham, A. Jabbari, J. S. Haring, and J. T. Harty. 2005. Accelerated CD8+ T-cell memory and prime-boost response after dendritic-cell vaccination. Nat. Med. 11:748-756. - PubMed
-
- Barber, D. L., E. J. Wherry, and R. Ahmed. 2003. Cutting edge: rapid in vivo killing by memory CD8 T cells. J. Immunol. 171:27-31. - PubMed
-
- Cameron, M. J., L. Ran, L. Xu, A. Danesh, J. F. Bermejo-Martin, C. M. Cameron, M. P. Muller, W. L. Gold, S. E. Richardson, S. M. Poutanen, B. M. Willey, M. E. DeVries, Y. Fang, C. Seneviratne, S. E. Bosinger, D. Persad, P. Wilkinson, L. D. Greller, R. Somogyi, A. Humar, S. Keshavjee, M. Louie, M. B. Loeb, J. Brunton, A. J. McGeer, S. R. N. Canadian, and D. J. Kelvin. 2007. Interferon-mediated immunopathological events are associated with atypical innate and adaptive immune responses in patients with severe acute respiratory syndrome. J. Virol. 81:8692-8706. - PMC - PubMed
-
- Chen, J., Y. F. Lau, E. W. Lamirande, C. D. Paddock, J. H. Bartlett, S. R. Zaki, and K. Subbarao. 2010. Cellular immune responses to severe acute respiratory syndrome coronavirus (SARS-CoV) infection in senescent BALB/c mice: CD4+ T cells are important in control of SARS-CoV infection. J. Virol. 84:1289-1301. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

