Topological basis for the robust distribution of blood to rodent neocortex
- PMID: 20616030
- PMCID: PMC2906564
- DOI: 10.1073/pnas.1007239107
Topological basis for the robust distribution of blood to rodent neocortex
Abstract
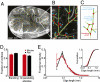
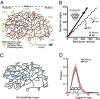
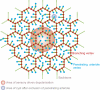
The maintenance of robust blood flow to the brain is crucial to the health of brain tissue. We examined the pial network of the middle cerebral artery, which distributes blood from the cerebral arteries to the penetrating arterioles that source neocortical microvasculature, to characterize how vascular topology may support such robustness. For both mice and rats, two features dominate the topology. First, interconnected loops span the entire territory sourced by the middle cerebral artery. Although the loops comprise <10% of all branches, they maintain the overall connectivity of the network after multiple breaks. Second, >80% of offshoots from the loops are stubs that end in a single penetrating arteriole, as opposed to trees with multiple penetrating arterioles. We hypothesize that the loops and stubs protect blood flow to the parenchyma from an occlusion in a surface vessel. To test this, we assayed the viability of tissue that was sourced by an individual penetrating arteriole following occlusion of a proximal branch in the surface loop. We observed that neurons remained healthy, even when occlusion led to a reduction in the local blood flow. In contrast, direct blockage of a single penetrating arteriole invariably led to neuronal death and formation of a cyst. Our results show that the surface vasculature functions as a grid for the robust allocation of blood in the event of vascular dysfunction. The combined results of the present and prior studies imply that the pial network reallocates blood in response to changing metabolic needs.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Schewe PF. The Grid: A Journey Through the Heart of Our Electrified World. Washington, DC: Joseph Henry Press; 2007.
-
- Albert R, Barabasi A-L. Statistical mechanics of complex networks. Rev Mod Phys. 2002;74:47–97.
-
- Banavar JR, Maritan A, Rinaldo A. Size and form in efficient transportation networks. Nature. 1999;399:130–132. - PubMed
-
- Bohn S, Magnasco MO. Structure, scaling, and phase transition in the optimal transport network. Phys Rev Lett. 2007;98:088702. - PubMed
-
- Thongpila S, et al. Adrenal microvascularization in the common tree shrew (Tupaia glis) as revealed by scanning electron microscopy of vascular corrosion casts. Acta Anat (Basel) 1998;163:31–38. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

