Structural basis for broad and potent neutralization of HIV-1 by antibody VRC01
- PMID: 20616231
- PMCID: PMC2981354
- DOI: 10.1126/science.1192819
Structural basis for broad and potent neutralization of HIV-1 by antibody VRC01
Abstract
During HIV-1 infection, antibodies are generated against the region of the viral gp120 envelope glycoprotein that binds CD4, the primary receptor for HIV-1. Among these antibodies, VRC01 achieves broad neutralization of diverse viral strains. We determined the crystal structure of VRC01 in complex with a human immunodeficiency virus HIV-1 gp120 core. VRC01 partially mimics CD4 interaction with gp120. A shift from the CD4-defined orientation, however, focuses VRC01 onto the vulnerable site of initial CD4 attachment, allowing it to overcome the glycan and conformational masking that diminishes the neutralization potency of most CD4-binding-site antibodies. To achieve this recognition, VRC01 contacts gp120 mainly through immunoglobulin V-gene regions substantially altered from their genomic precursors. Partial receptor mimicry and extensive affinity maturation thus facilitate neutralization of HIV-1 by natural human antibodies.
Figures




 ” marks a site of N-linked glycosylation, “
” marks a site of N-linked glycosylation, “ ” for cysteine residues involved in a non-canonical disulfide, and “
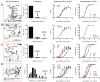
” for cysteine residues involved in a non-canonical disulfide, and “ ” if the residue has been deleted during affinity maturation. In B-E, unusual features of VRC01 are shown structurally (far left panel), in terms of frequency as a histogram with other antibodies (second panel from left), and in the context of affinity meaurements after mutational alteration (right two panels). Affinity measurements were made by ELISA to the gp120 construct used in crystallization (93TH057) and to a disulfide stabilized HXBc2 core (22). (B) N-linked glycosylation. The conserved tri-mannose core is shown with observed electron density, along with frequency and effect of removal on affinity. (C) Extra disulfide. Variable heavy domains naturally have two Cys, linked by a disulfide; VRC01 has an extra disulfide linking CDR H1 and H3 regions. This occurs rarely in antibodies, but its removal by mutation to Ser/Ala has little effect on affinity. (D) CDR L1 deletion. A two amino acid deletion in the CDR L1, prevents potential clashes with loop D of gp120. Such deletions are rarely observed; reversion to the longer loop may have a 10-100-fold effect on gp120 affinity. (E) Somatically altered contact surface. The far left panel shows the VRC01 light chain in violet and heavy chain in green. Residues altered by affinity maturation are depicted with “balls” and contacts with HIV-1 gp120 are colored red. About half the contacts are altered during the maturation process. Analysis of human antibody-protein complexes in the protein-data bank shows this degree of contact surface alteration is rare; reversion of each of the contact site to genome has little effect (Table S12), though in aggregate the effect on affinity is larger.
” if the residue has been deleted during affinity maturation. In B-E, unusual features of VRC01 are shown structurally (far left panel), in terms of frequency as a histogram with other antibodies (second panel from left), and in the context of affinity meaurements after mutational alteration (right two panels). Affinity measurements were made by ELISA to the gp120 construct used in crystallization (93TH057) and to a disulfide stabilized HXBc2 core (22). (B) N-linked glycosylation. The conserved tri-mannose core is shown with observed electron density, along with frequency and effect of removal on affinity. (C) Extra disulfide. Variable heavy domains naturally have two Cys, linked by a disulfide; VRC01 has an extra disulfide linking CDR H1 and H3 regions. This occurs rarely in antibodies, but its removal by mutation to Ser/Ala has little effect on affinity. (D) CDR L1 deletion. A two amino acid deletion in the CDR L1, prevents potential clashes with loop D of gp120. Such deletions are rarely observed; reversion to the longer loop may have a 10-100-fold effect on gp120 affinity. (E) Somatically altered contact surface. The far left panel shows the VRC01 light chain in violet and heavy chain in green. Residues altered by affinity maturation are depicted with “balls” and contacts with HIV-1 gp120 are colored red. About half the contacts are altered during the maturation process. Analysis of human antibody-protein complexes in the protein-data bank shows this degree of contact surface alteration is rare; reversion of each of the contact site to genome has little effect (Table S12), though in aggregate the effect on affinity is larger.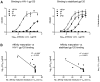
Comment in
-
AIDS/HIV. A boost for HIV vaccine design.Science. 2010 Aug 13;329(5993):770-3. doi: 10.1126/science.1194693. Science. 2010. PMID: 20705840 No abstract available.
References
-
- Weiss RA, et al. Nature. 1985 Jul 4-10;316:69.
-
- Mascola JR, Montefiori DC. Annu Rev Immunol. 2010 Mar;28:413. - PubMed
-
- Burton DR. Nat Rev Immunol. 2002 Sep;2:706. - PubMed
-
- Burton DR, et al. Nat Immunol. 2004 Mar;5:233. - PubMed
-
-
Antibodies 2F5 and 4E10 require non-specific membrane interaction (34-35), antibody 2G12 recognizes carbohydrate and is domain swapped (32-33), and antibody b12 was originally derived by phage display and has heavy-chain-only recognition (22).
-
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

