Insect cytokine paralytic peptide (PP) induces cellular and humoral immune responses in the silkworm Bombyx mori
- PMID: 20622022
- PMCID: PMC2937889
- DOI: 10.1074/jbc.M110.138446
Insect cytokine paralytic peptide (PP) induces cellular and humoral immune responses in the silkworm Bombyx mori
Abstract
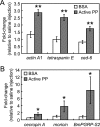
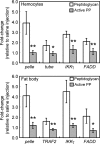
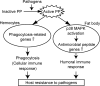
In the blood (hemolymph) of the silkworm Bombyx mori, the insect cytokine paralytic peptide (PP) is converted from an inactive precursor to an active form in response to the cell wall components of microorganisms and contributes to silkworm resistance to infection. To investigate the molecular mechanism underlying the up-regulation of host resistance induced by PP, we performed an oligonucleotide microarray analysis on RNA of blood cells (hemocytes) and fat body tissues of silkworm larvae injected with active PP. Expression levels of a large number of immune-related genes increased rapidly within 3 h after injecting active PP, including phagocytosis-related genes such as tetraspanin E, actin A1, and ced-6 in hemocytes, and antimicrobial peptide genes cecropin A and moricin in the fat body. Active PP promoted in vitro and in vivo phagocytosis of Staphyloccocus aureus by the hemocytes. Moreover, active PP induced in vivo phosphorylation of p38 mitogen-activated protein kinase (p38 MAPK) in the fat body. Pretreatment of silkworm larvae with ML3403, a pharmacologic p38 MAPK inhibitor, suppressed the PP-dependent induction of cecropin A and moricin genes in the fat body. Injection of active PP delayed the killing of silkworm larvae by S. aureus, whereas its effect was abolished by preinjection of the p38 MAPK inhibitor, suggesting that p38 MAPK activation is required for PP-dependent defensive responses. These findings suggest that PP acts on multiple tissues in silkworm larvae and acutely activates cellular and humoral immune responses, leading to host protection against infection.
Figures
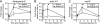



Similar articles
-
Paralytic peptide activates insect humoral immune response via epidermal growth factor receptor.Peptides. 2015 Sep;71:20-7. doi: 10.1016/j.peptides.2015.04.028. Epub 2015 May 20. Peptides. 2015. PMID: 26003397
-
Activation of the silkworm cytokine by bacterial and fungal cell wall components via a reactive oxygen species-triggered mechanism.J Biol Chem. 2008 Jan 25;283(4):2185-91. doi: 10.1074/jbc.M705480200. Epub 2007 Oct 18. J Biol Chem. 2008. PMID: 17947232
-
Insect cytokine paralytic peptide activates innate immunity via nitric oxide production in the silkworm Bombyx mori.Dev Comp Immunol. 2013 Mar;39(3):147-53. doi: 10.1016/j.dci.2012.10.014. Epub 2012 Nov 23. Dev Comp Immunol. 2013. PMID: 23178406
-
Haemocyte-mediated immunity in insects: Cells, processes and associated components in the fight against pathogens and parasites.Immunology. 2021 Nov;164(3):401-432. doi: 10.1111/imm.13390. Epub 2021 Aug 2. Immunology. 2021. PMID: 34233014 Free PMC article. Review.
-
Antimicrobial peptides from Bombyx mori: a splendid immune defense response in silkworms.RSC Adv. 2020 Jan 2;10(1):512-523. doi: 10.1039/c9ra06864c. eCollection 2019 Dec 20. RSC Adv. 2020. PMID: 35492565 Free PMC article. Review.
Cited by
-
Serine protease Bm-SP142 was differentially expressed in resistant and susceptible Bombyx mori strains, involving in the defence response to viral infection.PLoS One. 2017 Apr 17;12(4):e0175518. doi: 10.1371/journal.pone.0175518. eCollection 2017. PLoS One. 2017. PMID: 28414724 Free PMC article.
-
Symbiosis with Francisella tularensis provides resistance to pathogens in the silkworm.Sci Rep. 2016 Aug 10;6:31476. doi: 10.1038/srep31476. Sci Rep. 2016. PMID: 27507264 Free PMC article.
-
Thromboxane Mobilizes Insect Blood Cells to Infection Foci.Front Immunol. 2021 Dec 20;12:791319. doi: 10.3389/fimmu.2021.791319. eCollection 2021. Front Immunol. 2021. PMID: 34987515 Free PMC article.
-
Prediction of neuropeptide precursors and differential expression of adipokinetic hormone/corazonin-related peptide, hugin and corazonin in the brain of malaria vector Nyssorhynchus albimanus during a Plasmodium berghei infection.Curr Res Insect Sci. 2021 Apr 22;1:100014. doi: 10.1016/j.cris.2021.100014. eCollection 2021. Curr Res Insect Sci. 2021. PMID: 36003598 Free PMC article.
-
Serratia marcescens induces apoptotic cell death in host immune cells via a lipopolysaccharide- and flagella-dependent mechanism.J Biol Chem. 2012 Oct 19;287(43):36582-92. doi: 10.1074/jbc.M112.399667. Epub 2012 Aug 2. J Biol Chem. 2012. PMID: 22859304 Free PMC article.
References
-
- Hoffmann J. A., Reichhart J. M. (2002) Nat. Immunol. 3, 121–126 - PubMed
-
- Stuart L. M., Ezekowitz R. A. (2008) Nat. Rev. Immunol. 8, 131–141 - PubMed
-
- Boman H. G., Faye I., Gudmundsson G. H., Lee J. Y., Lidholm D. A. (1991) Eur. J. Biochem. 201, 23–31 - PubMed
-
- Natori S., Shiraishi H., Hori S., Kobayashi A. (1999) Dev. Comp. Immunol. 23, 317–328 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

