Control of HIF-1{alpha} and vascular signaling in fetal lung involves cross talk between mTORC1 and the FGF-10/FGFR2b/Spry2 airway branching periodicity clock
- PMID: 20622121
- PMCID: PMC2957420
- DOI: 10.1152/ajplung.00348.2009
Control of HIF-1{alpha} and vascular signaling in fetal lung involves cross talk between mTORC1 and the FGF-10/FGFR2b/Spry2 airway branching periodicity clock
Abstract
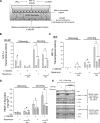
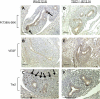
Lung development requires coordinated signaling between airway and vascular growth, but the link between these processes remains unclear. Mammalian target of rapamycin complex-1 (mTORC1) can amplify hypoxia-inducible factor-1α (HIF-1α) vasculogenic activity through an NH(2)-terminal mTOR binding (TOS) motif. We hypothesized that this mechanism coordinates vasculogenesis with the fibroblast growth factor (FGF)-10/FGF-receptor2b/Spry2 regulator of airway branching. First, we tested if the HIF-1α TOS motif participated in epithelial-mesenchymal vascular signaling. mTORC1 activation by insulin significantly amplified HIF-1α activity at fetal Po(2) (23 mmHg) in human bronchial epithelium (16HBE14o-) and induced vascular traits (Flk1, sprouting) in cocultured human embryonic lung mesenchyme (HEL-12469). This enhanced activation of HIF-1α by mTORC1 was abolished on expression of a HIF-1α (F99A) TOS-mutant and also suppressed vascular differentiation of HEL-12469 cocultures. Next, we determined if vasculogenesis in fetal lung involved regulation of mTORC1 by the FGF-10/FGFR2b/Spry2 pathway. Fetal airway epithelium displayed distinct mTORC1 activity in situ, and its hyperactivation by TSC1(-/-) knockout induced widespread VEGF expression and disaggregation of Tie2-positive vascular bundles. FGF-10-coated beads grafted into fetal lung explants from Tie2-LacZ transgenic mice induced localized vascular differentiation in the peripheral mesenchyme. In rat fetal distal lung epithelial (FDLE) cells cultured at fetal Po(2), FGF-10 induced mTORC1 and amplified HIF-1α activity and VEGF secretion without induction of ERK1/2. This was accompanied by the formation of a complex between Spry2, the cCBL ubiquitin ligase, and the mTOR repressor, TSC2, which abolished GTPase activity directed against Rheb, the G protein inducer of mTORC1. Thus, mTORC1 links HIF-1α-driven vasculogenesis with the FGF-10/FGFR2b/Spry2 airway branching periodicity regulator.
Figures










Comment in
-
Entrained pulmonary clocks: epithelium and vasculature keeping pace.Am J Physiol Lung Cell Mol Physiol. 2010 Oct;299(4):L453-4. doi: 10.1152/ajplung.00263.2010. Epub 2010 Aug 6. Am J Physiol Lung Cell Mol Physiol. 2010. PMID: 20693313 No abstract available.
References
-
- Acarregui MJ, Penisten ST, Goss KL, Ramirez K, Snyder JM. Vascular endothelial growth factor gene expression in human fetal lung in vitro. Am J Respir Cell Mol Biol 20: 14–23, 1999 - PubMed
-
- Asikainen TM, Chang LY, Coalson JJ, Schneider BK, Waleh NS, Ikegami M, Shannon JM, Winter VT, Grubb P, Clyman RI, Yoder BA, Crapo JD, White CW. Improved lung growth and function through hypoxia-inducible factor in primate chronic lung disease of prematurity. FASEB J 20: 1698–1700, 2006 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

