Aberrant striatal plasticity is specifically associated with dyskinesia following levodopa treatment
- PMID: 20623773
- PMCID: PMC3224800
- DOI: 10.1002/mds.23245
Aberrant striatal plasticity is specifically associated with dyskinesia following levodopa treatment
Abstract
Chronic levodopa treatment for Parkinson's disease often results in the development of abnormal involuntary movement, known as L-dopa-induced dyskinesia (LIDs). Studies suggest that LIDs may be associated with aberrant corticostriatal plasticity. Using in vivo extracellular recordings from identified Type I and Type II medium spiny striatal neurons, chronic L-dopa treatment was found to produce abnormal corticostriatal information processing. Specifically, after chronic L-dopa treatment in dopamine-depleted rats, there was a transition from a cortically evoked long-term depression (LTD) to a complementary but opposing form of plasticity, long-term potentiation, in Type II "indirect" pathway neurons. In contrast, LTD could still be induced in Type I neurons. Interestingly, the one parameter that correlated best with dyskinesias was the inability to de-depress established LTD in Type I medium spiny striatal neurons. Taken as a whole, we propose that the induction of LIDs is due, at least in part, to an aberrant induction of plasticity within the Type II indirect pathway neurons combined with an inability to de-depress established plastic responses in Type I neurons. Such information is critical for understanding the cellular mechanisms underlying one of the major caveats to L-dopa therapy.
Figures
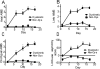
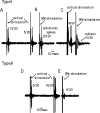
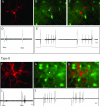


Comment in
-
Direct and indirect pathways in levodopa-induced dyskinesia: a more complex matter than a network imbalance.Mov Disord. 2010 Aug 15;25(11):1527-9. doi: 10.1002/mds.23244. Mov Disord. 2010. PMID: 20623774 No abstract available.
References
-
- Ehringer H, Hornykiewicz O. Distribution of noradrenaline and dopamine (3-hydroxytyramine) in the human brain and their behavior in diseases of the extrapyramidal system. Klin Wochenschr. 1960;38:1236–1239. - PubMed
-
- Marsden CD, Parkes JD. Success and problems of long-term levodopa therapy in Parkinson's disease. Lancet. 1977;1(8007):345–349. - PubMed
-
- Bedard PJ, Blanchet PJ, Levesque D, et al. Pathophysiology of L-dopa-induced dyskinesias. Mov Disord. 1999;14(Suppl 1):4–8. - PubMed
-
- Calabresi P, Giacomini P, Centonze D, Bernardi G. Levodopa-induced dyskinesia: a pathological form of striatal synaptic plasticity? Ann Neurol. 2000;47(4 Suppl 1):S60–68. discussion S68-69. - PubMed
-
- Chase TN, Oh JD. Striatal dopamine- and glutamate-mediated dysregulation in experimental parkinsonism. Trends Neurosci. 2000;23(10 Suppl):S86–91. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

