Intravenous anakinra can achieve experimentally effective concentrations in the central nervous system within a therapeutic time window: results of a dose-ranging study
- PMID: 20628399
- PMCID: PMC3049499
- DOI: 10.1038/jcbfm.2010.103
Intravenous anakinra can achieve experimentally effective concentrations in the central nervous system within a therapeutic time window: results of a dose-ranging study
Abstract
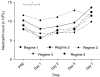
The naturally occurring antagonist of interleukin-1, IL-1RA, is highly neuroprotective experimentally, shows few adverse effects, and inhibits the systemic acute phase response to stroke. A single regime pilot study showed slow penetration into cerebrospinal fluid (CSF) at experimentally therapeutic concentrations. Twenty-five patients with subarachnoid hemorrhage (SAH) and external ventricular drains were sequentially allocated to five administration regimes, using intravenous bolus doses of 100 to 500 mg and 4 hours intravenous infusions of IL-1RA ranging from 1 to 10 mg per kg per hour. Choice of regimes and timing of plasma and CSF sampling was informed by pharmacometric analysis of pilot study data. Data were analyzed using nonlinear mixed effects modeling. Plasma and CSF concentrations of IL-1RA in all regimes were within the predicted intervals. A 500-mg bolus followed by an intravenous infusion of IL-1RA at 10 mg per kg per hour achieved experimentally therapeutic CSF concentrations of IL-1RA within 45 minutes. Experimentally, neuroprotective CSF concentrations in patients with SAH can be safely achieved within a therapeutic time window. Pharmacokinetic analysis suggests that IL-1RA transport across the blood-CSF barrier in SAH is passive. Identification of the practicality of this delivery regime allows further studies of efficacy of IL-1RA in acute cerebrovascular disease.
Figures


References
-
- Adams HP, Jr, del Zoppo G, Alberts MJ, Bhatt DL, Brass L, Furlan A, Grubb RL, Higashida RT, Jauch EC, Kidwell C, Lyden PD, Morgenstern LB, Qureshi AI, Rosenwasser RH, Scott PA, Wijdicks EF. Guidelines for the early management of adults with ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: The American Academy of Neurology affirms the value of this guideline as an educational tool for neurologists. Circulation. 2007;115:e478–e534. - PubMed
-
- Arabi Y, Memish ZA, Balkhy HH, Francis C, Ferayan A, Al Shimemeri A, Almuneef MA. Ventriculostomy-associated infections: incidence and risk factors. Am J Infect Control. 2005;33:137–143. - PubMed
-
- Banks WA, Ortiz L, Plotkin SR, Kastin AJ. Human interleukin (IL) 1 alpha, murine IL-1 alpha and murine IL-1 beta are transported from blood to brain in the mouse by a shared saturable mechanism. J Pharmacol Exp Ther. 1991;259:988–996. - PubMed
-
- Banwell V, Sena ES, Macleod MR. Systematic review and stratified meta-analysis of the efficacy of interleukin-1 receptor antagonist in animal models of stroke. J Stroke Cerebrovasc Dis. 2009;18:269–276. - PubMed
-
- Berrouane Y, Daudenthun I, Riegel B, Emery MN, Martin G, Krivosic R, Grandbastien B. Early onset pneumonia in neurosurgical intensive care unit patients. J Hosp Infect. 1998;40:275–280. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

