Enhanced receptor expression and in vitro effector function of a murine-human hybrid MART-1-reactive T cell receptor following a rapid expansion
- PMID: 20628878
- PMCID: PMC3436071
- DOI: 10.1007/s00262-010-0882-5
Enhanced receptor expression and in vitro effector function of a murine-human hybrid MART-1-reactive T cell receptor following a rapid expansion
Abstract
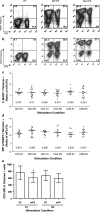
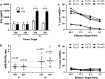
Peripheral blood lymphocytes (PBL) genetically modified to express T cell receptors (TCR) specific to known melanoma antigens, such as melanoma antigen recognized by T cells-1 (MART-1), and gp100 can elicit objective tumor regression when administered to patients with metastatic melanoma. It has also been demonstrated that modifications within the constant regions of a fully human TCR can enhance surface expression and stability without altering antigen specificity. In this study, we evaluated the substitution of murine constant regions for their human counterpart within the DMF5 MART-1-specific TCR. Unlike previous studies, all modified TCRs were inserted into retroviral vectors and analyzed for expression and function following a clinical transduction protocol. PBL were transduced with retroviral supernatant generated from stable packaging lines encoding melanoma-specific TCRs. This protocol resulted in high levels of antigen-specific T cells without the need for additional peptide stimulation and selection. Both the human and murinized TCR efficiently transduced PBL; however, the murinized TCR exhibited significantly higher tetramer binding, mean fluorescence intensity, as well as, increased in vitro effector function following our clinical transduction and expansion protocol. Additional TCR modifications including insertion of a second disulfide bond or the linker modifications evaluated herein did not significantly enhance TCR expression or subsequent in vitro effector function. We conclude that the substitution of a human constant region with a murine constant region was sufficient to increase receptor expression and tetramer binding as well as antitumor activity of the DMF5 TCR and could be a tool to augment other antigen-specific TCR.
Figures



References
-
- Balch C, Atkins M, Sober A. Cutaneous Melanoma. In: Devita V Jr, Helman S, Rosenber S, editors. Cancer: principles and practice of oncology. 7. Philadelphia: Lippincott Willliams & Wilkins; 2005. pp. 1754–1808.
-
- Dudley ME, Wunderlich JR, Yang JC, Sherry RM, Topalian SL, et al. Adoptive cell transfer therapy following non-myeloablative but lymphodepleting chemotherapy for the treatment of patients with refractory metastatic melanoma. J Clin Oncol. 2005;23:2346–2357. doi: 10.1200/JCO.2005.00.240. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

