Regeneration of the MPTP-lesioned dopaminergic system after convection-enhanced delivery of AAV2-GDNF
- PMID: 20631185
- PMCID: PMC2914692
- DOI: 10.1523/JNEUROSCI.0942-10.2010
Regeneration of the MPTP-lesioned dopaminergic system after convection-enhanced delivery of AAV2-GDNF
Abstract
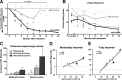
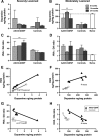
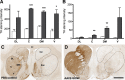
Clinical studies to date have failed to establish therapeutic benefit of glial cell-derived neurotrophic factor (GDNF) in Parkinson's disease (PD). In contrast to previous nonclinical neuroprotective reports, this study shows clinically relevant and long-lasting regeneration of the dopaminergic system in rhesus macaques lesioned with 1-methy-4-phenyl-1,2,3,6-tetrahydropyridine 3-6 months before GDNF gene delivery (AAV2-GDNF). The observed progressive amelioration of functional deficits, recovery of dopamine, and regrowth of fibers to the striatal neuropil demonstrate that high GDNF expression in the putamen promotes restoration of the dopaminergic system in a primate model of advanced PD. Extensive distribution of GDNF within the putamen and transport to the severely lesioned substantia nigra, after convection-enhanced delivery of AAV2-GDNF into the putamen, indicates anterograde transport via striatonigral connections and is anticipated to occur in PD patients. Overall, these data demonstrate nonclinical neurorestoration after putaminal infusion of AAV2-GDNF and suggest that clinical investigation in PD patients is warranted.
Figures


Similar articles
-
Clinically relevant effects of convection-enhanced delivery of AAV2-GDNF on the dopaminergic nigrostriatal pathway in aged rhesus monkeys.Hum Gene Ther. 2009 May;20(5):497-510. doi: 10.1089/hum.2008.137. Hum Gene Ther. 2009. PMID: 19203243 Free PMC article.
-
Safety evaluation of AAV2-GDNF gene transfer into the dopaminergic nigrostriatal pathway in aged and parkinsonian rhesus monkeys.Hum Gene Ther. 2009 Dec;20(12):1627-40. doi: 10.1089/hum.2009.103. Hum Gene Ther. 2009. PMID: 19671001 Free PMC article.
-
Neurodegeneration prevented by lentiviral vector delivery of GDNF in primate models of Parkinson's disease.Science. 2000 Oct 27;290(5492):767-73. doi: 10.1126/science.290.5492.767. Science. 2000. PMID: 11052933
-
Towards a neuroprotective gene therapy for Parkinson's disease: use of adenovirus, AAV and lentivirus vectors for gene transfer of GDNF to the nigrostriatal system in the rat Parkinson model.Brain Res. 2000 Dec 15;886(1-2):82-98. doi: 10.1016/s0006-8993(00)02915-2. Brain Res. 2000. PMID: 11119690 Review.
-
Adenoviral vector-mediated delivery of glial cell line-derived neurotrophic factor provides neuroprotection in the aged parkinsonian rat.Clin Exp Pharmacol Physiol. 2001 Nov;28(11):896-900. doi: 10.1046/j.1440-1681.2001.03544.x. Clin Exp Pharmacol Physiol. 2001. PMID: 11703392 Review.
Cited by
-
Anterograde axonal transport of AAV2-GDNF in rat basal ganglia.Mol Ther. 2011 May;19(5):922-7. doi: 10.1038/mt.2010.248. Epub 2010 Nov 23. Mol Ther. 2011. PMID: 21102559 Free PMC article.
-
Comparative Analysis of the Effects of Neurotrophic Factors CDNF and GDNF in a Nonhuman Primate Model of Parkinson's Disease.PLoS One. 2016 Feb 22;11(2):e0149776. doi: 10.1371/journal.pone.0149776. eCollection 2016. PLoS One. 2016. PMID: 26901822 Free PMC article.
-
Adult Endogenous Dopaminergic Neuroregeneration Against Parkinson's Disease: Ideal Animal Models?Neurotox Res. 2021 Apr;39(2):504-532. doi: 10.1007/s12640-020-00298-7. Epub 2020 Nov 3. Neurotox Res. 2021. PMID: 33141428 Review.
-
Intraputaminal Delivery of Adeno-Associated Virus Serotype 2-Glial Cell Line-Derived Neurotrophic Factor in Mild or Moderate Parkinson's Disease.Mov Disord. 2025 Jul;40(7):1297-1306. doi: 10.1002/mds.30193. Epub 2025 May 20. Mov Disord. 2025. PMID: 40395017 Free PMC article. Clinical Trial.
-
Adeno-associated virus (AAV) gene therapy for neurological disease.Neuropharmacology. 2013 Jun;69:82-8. doi: 10.1016/j.neuropharm.2012.03.004. Epub 2012 Mar 17. Neuropharmacology. 2013. PMID: 22465202 Free PMC article. Review.
References
-
- Bankiewicz KS, Oldfield EH, Chiueh CC, Doppman JL, Jacobowitz DM, Kopin IJ. Hemiparkinsonism in monkeys after unilateral internal carotid artery infusion of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) Life Sci. 1986;39:7–16. - PubMed
-
- Bankiewicz KS, Eberling JL, Kohutnicka M, Jagust W, Pivirotto P, Bringas J, Cunningham J, Budinger TF, Harvey-White J. Convection-enhanced delivery of AAV vector in parkinsonian monkeys: in vivo detection of gene expression and restoration of dopaminergic function using pro-drug approach. Exp Neurol. 2000;164:2–14. - PubMed
-
- Bankiewicz KS, Sanchez-Pernaute R, Oiwa Y, Kohutnicka M, Cummins A, Eberling J. Preclinical models of Parkinson's disease. Curr Protoc Neurosci. 2001 Chapter 9:Unit 9.4. - PubMed
-
- Bankiewicz KS, Forsayeth J, Eberling JL, Sanchez-Pernaute R, Pivirotto P, Bringas J, Herscovitch P, Carson RE, Eckelman W, Reutter B, Cunningham J. Long-term clinical improvement in MPTP-lesioned primates after gene therapy with AAV-hAADC. Mol Ther. 2006;14:564–570. - PubMed
-
- Ceregene, Inc. Ceregene announces clinical data from phase 2 clinical trial of CERE-120 for Parkinson's disease. 2008. Press release, Nov. 26, 2008. Retrieved November 26, 2008.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
