Far-red fluorescent protein excitable with red lasers for flow cytometry and superresolution STED nanoscopy
- PMID: 20643047
- PMCID: PMC2905082
- DOI: 10.1016/j.bpj.2010.04.025
Far-red fluorescent protein excitable with red lasers for flow cytometry and superresolution STED nanoscopy
Abstract
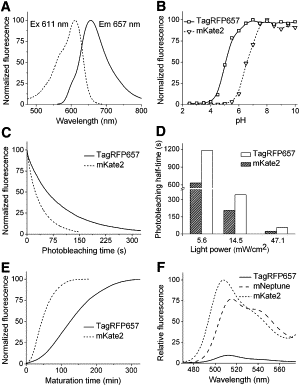
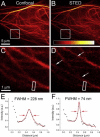
Far-red fluorescent proteins are required for deep-tissue and whole-animal imaging and multicolor labeling in the red wavelength range, as well as probes excitable with standard red lasers in flow cytometry and fluorescence microscopy. Rapidly evolving superresolution microscopy based on the stimulated emission depletion approach also demands genetically encoded monomeric probes to tag intracellular proteins at the molecular level. Based on the monomeric mKate variant, we have developed a far-red TagRFP657 protein with excitation/emission maxima at 611/657 nm. TagRFP657 has several advantages over existing monomeric far-red proteins including higher photostability, better pH stability, lower residual green fluorescence, and greater efficiency of excitation with red lasers. The red-shifted excitation and emission spectra, as compared to other far-red proteins, allows utilizing TagRFP657 in flow cytometry and fluorescence microscopy simultaneously with orange or near-red fluorescence proteins. TagRFP657 is shown to be an efficient protein tag for the superresolution fluorescence imaging using a commercially available stimulated emission depletion microscope.
Copyright (c) 2010 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

