Protein determinants of SNARE-mediated lipid mixing
- PMID: 20643074
- PMCID: PMC2905075
- DOI: 10.1016/j.bpj.2010.04.060
Protein determinants of SNARE-mediated lipid mixing
Abstract
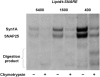
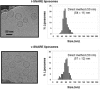
Soluble N-ethylmaleimide sensitive factor attachment protein receptor (SNARE)-mediated lipid mixing can be efficiently recapitulated in vitro by the incorporation of purified vesicle membrane (-v) SNARE and target membrane (t-) SNARE proteins into separate liposome populations. Despite the strong correlation between the observed activities in this system and the known SNARE physiology, some recent works have suggested that SNARE-mediated lipid mixing may be limited to circumstances where membrane defects arise from artifactual reconstitution conditions (such as nonphysiological high-protein concentrations or unrealistically small liposome populations). Here, we show that the previously published strategies used to reconstitute SNAREs into liposomes do not significantly affect either the physical parameters of the proteoliposomes or the ability of SNAREs to drive lipid mixing in vitro. The surface density of SNARE proteins turns out to be the most critical parameter, which controls both the rate and the extent of SNARE-mediated liposome fusion. In addition, the specific activity of the t-SNARE complex is significantly influenced by expression and reconstitution protocols, such that we only observe optimal lipid mixing when the t-SNARE proteins are coexpressed before purification.
Copyright (c) 2010 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures



References
-
- Söllner T., Whiteheart S.W., Rothman J.E. SNAP receptors implicated in vesicle targeting and fusion. Nature. 1993;362:318–324. - PubMed
-
- Brunger A.T. Structure and function of SNARE and SNARE-interacting proteins. Q. Rev. Biophys. 2005;38:1–47. - PubMed
-
- Jackson M.B., Chapman E.R. Fusion pores and fusion machines in Ca2+-triggered exocytosis. Annu. Rev. Biophys. Biomol. Struct. 2006;35:135–160. - PubMed
-
- Jahn R., Scheller R.H. SNAREs—engines for membrane fusion. Nat. Rev. Mol. Cell Biol. 2006;7:631–643. - PubMed
-
- Hanson P.I., Roth R., Heuser J.E. Structure and conformational changes in NSF and its membrane receptor complexes visualized by quick-freeze/deep-etch electron microscopy. Cell. 1997;90:523–535. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

