Trabectedin plus pegylated liposomal doxorubicin in relapsed ovarian cancer delays third-line chemotherapy and prolongs the platinum-free interval
- PMID: 20643863
- PMCID: PMC3003617
- DOI: 10.1093/annonc/mdq353
Trabectedin plus pegylated liposomal doxorubicin in relapsed ovarian cancer delays third-line chemotherapy and prolongs the platinum-free interval
Abstract
Background: OVA-301 is a large randomized trial that showed superiority of trabectedin plus pegylated liposomal doxorubicin (PLD; CentoCor Ortho Biotech Products L.P., Raritan, NJ, USA). over single-agent PLD in 672 patients with relapsed ovarian cancer, particularly in the partially platinum-sensitive subgroup [platinum-free interval (PFI) of 6-12 months]. This superiority has been suggested to be due to the differential impact of subsequent (platinum) therapy.
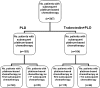
Patients and methods: a detailed analysis of subsequent therapies and survival outcomes in the overall population and in the subsets according to platinum sensitivity was therefore conducted.
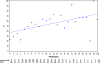
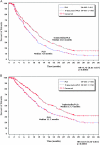
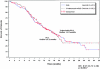
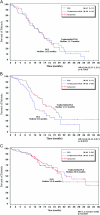
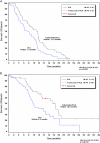
Results: similar proportions of patients received subsequent therapy in each arm (76% versus 77%), including further platinum-based regimens (49% versus 55%). Patients in the trabectedin/PLD arm received subsequent chemotherapy at a later time (median delay 2.5 months versus PLD arm). Overall survival from subsequent platinum was significantly prolonged in the partially platinum-sensitive disease subset (hazard ratio = 0.63; P = 0.0357).
Conclusion: the superiority of trabectedin/PLD over single-agent PLD in OVA-301 cannot be explained by differences in the extent or nature of subsequent therapies administered to these patients. On the other hand, these exploratory analyses support the hypothesis that the enhanced survival benefits in the partially platinum-sensitive subset might be due to an extended PFI leading to longer survival with subsequent platinum.
Figures
Comment in
-
Trabectedin in ovarian cancer: could we expect more?Ann Oncol. 2011 Jan;22(1):7-8. doi: 10.1093/annonc/mdq641. Epub 2010 Nov 8. Ann Oncol. 2011. PMID: 21059638 No abstract available.
References
-
- Demetri GD, Chawla SP, von Mehren M, et al. Efficacy and safety of trabectedin in patients with advanced or metastatic liposarcoma or leiomyosarcoma after failure of prior anthracyclines and ifosfamide: results of a randomized phase II study of two different schedules. J Clin Oncol. 2009;27:4188–4196. - PubMed
-
- Carter NJ, Keam SJ. Trabectedin: a review of its use in the management of soft tissue sarcoma and ovarian cancer. Drugs. 2007;67:2257–2276. - PubMed
-
- Ganjoo KN, Patel SR. Trabectedin: an anticancer drug from the sea. Expert Opin Pharmacother. 2009;10:2735–2743. - PubMed
-
- Le Cesne A, Domont J, Cioffi A, et al. Mapping the literature: role of trabectedin as a new chemotherapy option in advanced pretreated soft tissue sarcoma. Drugs Today (Barc) 2009;45:403–421. - PubMed
-
- Sessa C, De Braud F, Perotti A, et al. Trabectedin for women with ovarian carcinoma after treatment with platinum and taxanes fails. J Clin Oncol. 2005;23:1867–1874. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

