The expression profile of the major mouse SPO11 isoforms indicates that SPO11beta introduces double strand breaks and suggests that SPO11alpha has an additional role in prophase in both spermatocytes and oocytes
- PMID: 20647542
- PMCID: PMC2937527
- DOI: 10.1128/MCB.00002-10
The expression profile of the major mouse SPO11 isoforms indicates that SPO11beta introduces double strand breaks and suggests that SPO11alpha has an additional role in prophase in both spermatocytes and oocytes
Abstract
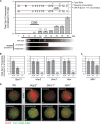
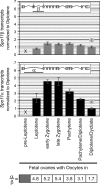
Both in mice and humans, two major SPO11 isoforms are generated by alternative splicing: SPO11alpha (exon 2 skipped) and SPO11beta. Thus, the alternative splicing event must have emerged before the mouse and human lineages diverged and was maintained during 90 million years of evolution, arguing for an essential role for both isoforms. Here we demonstrate that developmental regulation of alternative splicing at the Spo11 locus governs the sequential expression of SPO11 isoforms in male meiotic prophase. Protein quantification in juvenile mice and in prophase mutants indicates that early spermatocytes synthesize primarily SPO11beta. Estimation of the number of SPO11 dimers (betabeta/alphabeta/alphaalpha) in mutants in which spermatocytes undergo a normal number of double strand breaks but arrest in midprophase due to inefficient repair argues for a role for SPO11beta-containing dimers in introducing the breaks in leptonema. Expression kinetics in males suggested a role for SPO11alpha in pachytene/diplotene spermatocytes. Nevertheless, we found that both alternative transcripts can be detected in oocytes throughout prophase I, arguing against a male-specific function for this isoform. Altogether, our data support a role for SPO11alpha in mid- to late prophase, presumably acting as a topoisomerase, that would be conserved in male and female meiocytes.
Figures




References
-
- Baker, S. M., A. W. Plug, T. A. Prolla, C. E. Bronner, A. C. Harris, X. Yao, D. M. Christie, C. Monell, N. Arnheim, A. Bradley, T. Ashley, and R. M. Liskay. 1996. Involvement of mouse Mlh1 in DNA mismatch repair and meiotic crossing over. Nat. Genet. 13:336-342. - PubMed
-
- Barchi, M., S. Mahadevaiah, M. Di Giacomo, F. Baudat, D. G. de Rooij, P. S. Burgoyne, M. Jasin, and S. Keeney. 2005. Surveillance of different recombination defects in mouse spermatocytes yields distinct responses despite elimination at an identical developmental stage. Mol. Cell. Biol. 25:7203-7215. - PMC - PubMed
-
- Barlow, C., S. Hirotsune, R. Paylor, M. Liyanage, M. Eckhaus, F. Collins, Y. Shiloh, J. N. Crawley, T. Ried, D. Tagle, and A. Wynshaw-Boris. 1996. Atm-deficient mice: a paradigm of ataxia telangiectasia. Cell 86:159-171. - PubMed
-
- Barlow, C., M. Liyanage, P. B. Moens, M. Tarsounas, K. Nagashima, K. Brown, S. Rottinghaus, S. P. Jackson, D. Tagle, T. Ried, and A. Wynshaw-Boris. 1998. Atm deficiency results in severe meiotic disruption as early as leptonema of prophase I. Development 125:4007-4017. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
