Neurosteroid transport by the organic solute transporter OSTα-OSTβ
- PMID: 20649839
- PMCID: PMC2939961
- DOI: 10.1111/j.1471-4159.2010.06920.x
Neurosteroid transport by the organic solute transporter OSTα-OSTβ
Abstract
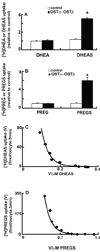
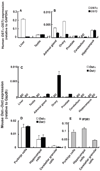
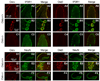
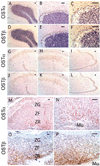
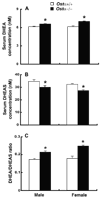
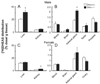
A variety of steroids, including pregnenolone sulfate (PREGS) and dehydroepiandrosterone sulfate (DHEAS) are synthesized by specific brain cells, and are then delivered to their target sites, where they exert potent effects on neuronal excitability. The present results demonstrate that [(3)H]DHEAS and [(3)H]PREGS are relatively high affinity substrates for the organic solute transporter, OSTα-OSTβ, and that the two proteins that constitute this transporter are selectively localized to steroidogenic cells in the cerebellum and hippocampus, namely the Purkinje cells and cells in the cornu ammonis region in both mouse and human brain. Analysis of Ostα and Ostβ mRNA levels in mouse Purkinje and hippocampal cells isolated via laser capture microdissection supported these findings. In addition, Ostα-deficient mice exhibited changes in serum DHEA and DHEAS levels, and in tissue distribution of administered [(3)H]DHEAS. OSTα and OSTβ proteins were also localized to the zona reticularis of human adrenal gland, the major region for DHEAS production in the periphery. These results demonstrate that OSTα-OSTβ is localized to steroidogenic cells of the brain and adrenal gland, and that it modulates DHEA/DHEAS homeostasis, suggesting that it may contribute to neurosteroid action.
© 2010 The Authors. Journal Compilation © 2010 International Society for Neurochemistry.
Conflict of interest statement
The authors affirm that there are no conflicts of interest.
Figures


References
-
- Aoki T, Narita M, Ohnishi O, Mizuo K, Narita M, Yajima Y, Suzuki T. Disruption of the type 1 inositol 1,4,5-trisphosphate receptor gene suppresses the morphine-induced antinociception in the mouse. Neurosci.. Lett. 2003;350:69–72. - PubMed
-
- Asif AR, Steffgen J, Metten M, Grunewald RW, Müller GA, Bahn A, Burckhardt G, Hagos Y. Presence of organic anion transporters 3 (OAT3) and 4 (OAT4) in human adrenocortical cells. Pflugers Arch. 2005;450:88–95. - PubMed
-
- Ballatori N, Christian WV, Lee JY, Dawson PA, Soroka CJ, Boyer JL, Madejczyk MS, Li N. OSTα-OSTβ: a major basolateral bile acid and steroid transporter in human intestinal, renal, and biliary epithelia. Hepatology. 2005;42:1270–1279. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R21 ES017470/ES/NIEHS NIH HHS/United States
- ES01247/ES/NIEHS NIH HHS/United States
- P30 ES001247/ES/NIEHS NIH HHS/United States
- P01 CA095616/CA/NCI NIH HHS/United States
- ES014899/ES/NIEHS NIH HHS/United States
- U24 NS050606/NS/NINDS NIH HHS/United States
- ES07026/ES/NIEHS NIH HHS/United States
- R01 ES014899/ES/NIEHS NIH HHS/United States
- U24NS050606/NS/NINDS NIH HHS/United States
- DK067214/DK/NIDDK NIH HHS/United States
- R01 DK067214/DK/NIDDK NIH HHS/United States
- T32 ES007026/ES/NIEHS NIH HHS/United States
- R01 ES007026/ES/NIEHS NIH HHS/United States
- ES17470/ES/NIEHS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

