A new mode of mineralocorticoid receptor antagonism by a potent and selective nonsteroidal molecule
- PMID: 20650892
- PMCID: PMC2943305
- DOI: 10.1074/jbc.M110.131342
A new mode of mineralocorticoid receptor antagonism by a potent and selective nonsteroidal molecule
Abstract
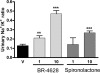
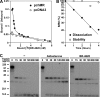
Limitations of current steroidal mineralocorticoid receptor (MR) antagonists have stimulated the search for a new generation of molecules. We screened for novel nonsteroidal compounds and identified MR antagonists derived from the chemical class of dihydropyridines. Chemical optimization resulted in BR-4628, which displays high in vitro and in vivo MR potency as well as selectivity with respect to the other steroid hormone receptors and the L-type calcium channel. Biochemical studies demonstrated that BR-4628 forms complexes with MR that do not promote the recruitment of transcriptional co-regulators. Docking experiments, using the crystal structure of the MR ligand-binding domain in an agonist conformation, revealed that BR-4628 accommodates in the MR ligand-binding cavity differently in comparison with the classical steroidal MR antagonists. An alanine scanning mutagenesis approach, based on BR-4628 docking, allowed identifying its anchoring mode within the ligand-binding cavity. Altogether, we propose that BR-4628 is a bulky antagonist that inactivates MR through a passive mechanism. It represents the prototype of a new class of MR antagonists.
Figures

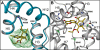

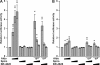
References
-
- Horisberger J. D., Rossier B. C. (1992) Hypertension 19, 221–227 - PubMed
-
- Bonvalet J. P. (1998) Kidney Int. Suppl. 65, S49–S56 - PubMed
-
- Geller D. S., Rodriguez-Soriano J., Vallo Boado A., Schifter S., Bayer M., Chang S. S., Lifton R. P. (1998) Nat. Genet. 19, 279–281 - PubMed
-
- Sartorato P., Cluzeaud F., Fagart J., Viengchareun S., Lombès M., Zennaro M. C. (2004) Mol. Endocrinol. 18, 2151–2165 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

