A double-blind, randomized, placebo and active-controlled study of nebicapone for the treatment of motor fluctuations in Parkinson's disease
- PMID: 20653695
- PMCID: PMC6493781
- DOI: 10.1111/j.1755-5949.2010.00145.x
A double-blind, randomized, placebo and active-controlled study of nebicapone for the treatment of motor fluctuations in Parkinson's disease
Abstract
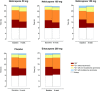
To determine the efficacy, safety and tolerability of nebicapone, a new catechol-O-methyltransferase inhibitor for the treatment of motor fluctuations in Parkinson's disease (PD), we conducted a multicenter, randomized, 8-week double-blind, placebo- and active-controlled, parallel-group study comparing nebicapone 50 mg, 100 mg, or 150 mg, entacapone 200 mg (active control) or placebo administered concomitantly with levodopa/carbidopa or levodopa/benserazide. Two hundred and fifty-two PD patients with motor fluctuations treated with levodopa/carbidopa or levodopa/benserazide (4-8 daily doses) were enrolled and 250 patients were eligible for intention-to-treat (ITT) analysis on the basis of having at least one efficacy assessment. The primary endpoint was 8-week change from baseline in absolute "Off" time duration noted in self-scoring diaries. At 8 weeks of treatment the mean daily "Off" time decreased significantly compared to placebo for nebicapone 150 mg (-106 min; 95%CI: -192; -21) and entacapone 200 mg (-81 min; 95%CI: -142; -19). The decrease in "Off" time with nebicapone 50 mg or 100 mg did not reach statistical significance. Treatment-emergent adverse events were reported by 32% to 49% of patients in any treatment group, with no observed dose relationship in the nebicapone groups. Clinically relevant elevations in aspartate transaminase (AST) and/or alanine transaminase (ALT) were observed in 4 of 46 patients with the nebicapone 150 mg dose. The results of this study show that nebicapone 150 mg is efficacious for the treatment of motor fluctuations in PD patients. However, the risk of increasing liver transaminases and its clinically relevance deserves further evaluation.
© 2010 Blackwell Publishing Ltd.
Conflict of interest statement
The authors have no conflict of interest.
Figures
References
-
- Lewitt PA. Levodopa for the treatment of Parkinson's disease. N Engl J Med 2008;359:2468–2476. - PubMed
-
- Olanow CW, Agid Y, Mizuno Y, et al Levodopa in the treatment of Parkinson's disease: Current controversies. Mov Disord 2004;19:997–1005. - PubMed
-
- Jankovic J. Motor fluctuations and dyskinesias in Parkinson's disease: Clinical manifestations. Mov Disord 2005;20(Suppl 11):S11–S16. - PubMed
-
- Schrag A, Quinn N. Dyskinesias and motor fluctuations in Parkinson's disease. A community‐based study. Brain 2000;123(Pt 11):2297–2305. - PubMed
-
- Goetz CG, Poewe W, Rascol O, Sampaio C. Evidencebased medical review update: Pharmacological and surgical treatments of Parkinson's disease: 2001 to 2004. Mov Disord 2005;20:523–539. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical