The homeobox transcription factor Barx2 regulates plasticity of young primary myofibers
- PMID: 20657655
- PMCID: PMC2904708
- DOI: 10.1371/journal.pone.0011612
The homeobox transcription factor Barx2 regulates plasticity of young primary myofibers
Abstract
Background: Adult mammalian muscle retains incredible plasticity. Muscle growth and repair involves the activation of undifferentiated myogenic precursors called satellite cells. In some circumstances, it has been proposed that existing myofibers may also cleave and produce a pool of proliferative cells that can re-differentiate into new fibers. Such myofiber dedifferentiation has been observed in the salamander blastema where it may occur in parallel with satellite cell activation. Moreover, ectopic expression of the homeodomain transcription factor Msx1 in differentiated C2C12 myotubes has been shown to induce their dedifferentiation. While it remains unclear whether dedifferentiation and redifferentiaton occurs endogenously in mammalian muscle, there is considerable interest in induced dedifferentiation as a possible regenerative tool.
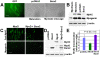
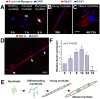
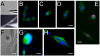
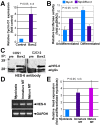
Methodology/principal findings: We previously showed that the homeobox protein Barx2 promotes myoblast differentiation. Here we report that ectopic expression of Barx2 in young immature myotubes derived from cell lines and primary mouse myoblasts, caused cleavage of the syncytium and downregulation of differentiation markers. Microinjection of Barx2 cDNA into immature myotubes derived from primary cells led to cleavage and formation of mononucleated cells that were able to proliferate. However, injection of Barx2 cDNA into mature myotubes did not cause cleavage. Barx2 expression in C2C12 myotubes increased the expression of cyclin D1, which may promote cell cycle re-entry. We also observed differential muscle gene regulation by Barx2 at early and late stages of muscle differentiation which may be due to differential recruitment of transcriptional activator or repressor complexes to muscle specific genes by Barx2.
Conclusions/significance: We show that Barx2 regulates plasticity of immature myofibers and might act as a molecular switch controlling cell differentiation and proliferation.
Conflict of interest statement
Figures
Similar articles
-
Barx2 and Pax7 have antagonistic functions in regulation of wnt signaling and satellite cell differentiation.Stem Cells. 2014 Jun;32(6):1661-73. doi: 10.1002/stem.1674. Stem Cells. 2014. PMID: 24753152 Free PMC article.
-
The homeodomain protein Barx2 promotes myogenic differentiation and is regulated by myogenic regulatory factors.J Biol Chem. 2003 Mar 7;278(10):8269-78. doi: 10.1074/jbc.M207617200. Epub 2002 Dec 16. J Biol Chem. 2003. PMID: 12486129
-
Dedifferentiation of mammalian myotubes induced by msx1.Cell. 2000 Dec 22;103(7):1099-109. doi: 10.1016/s0092-8674(00)00212-9. Cell. 2000. PMID: 11163185
-
Barx homeobox family in muscle development and regeneration.Int Rev Cell Mol Biol. 2012;297:117-73. doi: 10.1016/B978-0-12-394308-8.00004-2. Int Rev Cell Mol Biol. 2012. PMID: 22608559 Review.
-
Restoring the Cell Cycle and Proliferation Competence in Terminally Differentiated Skeletal Muscle Myotubes.Cells. 2021 Oct 14;10(10):2753. doi: 10.3390/cells10102753. Cells. 2021. PMID: 34685732 Free PMC article. Review.
Cited by
-
Establishment of a Lymph Node Metastasis-Associated Prognostic Signature for Lung Adenocarcinoma.Genet Res (Camb). 2023 Jan 31;2023:6585109. doi: 10.1155/2023/6585109. eCollection 2023. Genet Res (Camb). 2023. PMID: 36793937 Free PMC article.
-
Satellite cells: regenerative mechanisms and applicability in muscular dystrophy.Stem Cells Int. 2015;2015:487467. doi: 10.1155/2015/487467. Epub 2015 Feb 11. Stem Cells Int. 2015. PMID: 25763072 Free PMC article. Review.
-
Down-regulation of myogenin can reverse terminal muscle cell differentiation.PLoS One. 2012;7(1):e29896. doi: 10.1371/journal.pone.0029896. Epub 2012 Jan 3. PLoS One. 2012. PMID: 22235349 Free PMC article.
-
Satellite cells and the muscle stem cell niche.Physiol Rev. 2013 Jan;93(1):23-67. doi: 10.1152/physrev.00043.2011. Physiol Rev. 2013. PMID: 23303905 Free PMC article. Review.
-
Thimerosal-induced apoptosis in mouse C2C12 myoblast cells occurs through suppression of the PI3K/Akt/survivin pathway.PLoS One. 2012;7(11):e49064. doi: 10.1371/journal.pone.0049064. Epub 2012 Nov 7. PLoS One. 2012. PMID: 23145070 Free PMC article.
References
-
- Buckingham M. Skeletal muscle progenitor cells and the role of Pax genes. Comptes Rendus Biologies. 2007;330:530–533. - PubMed
-
- Campion DR. The muscle satellite cell: a review. Int Rev Cytol. 1984;87:225–251. - PubMed
-
- Rudnicki MA, Le Grand F, McKinnell I, Kuang S. The molecular regulation of muscle stem cell function. Cold Spring Harb Symp Quant Biol. 2008;73:323–331. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

