Importance of non-selective cation channel TRPV4 interaction with cytoskeleton and their reciprocal regulations in cultured cells
- PMID: 20657843
- PMCID: PMC2906515
- DOI: 10.1371/journal.pone.0011654
Importance of non-selective cation channel TRPV4 interaction with cytoskeleton and their reciprocal regulations in cultured cells
Abstract
Background: TRPV4 and the cellular cytoskeleton have each been reported to influence cellular mechanosensitive processes as well as the development of mechanical hyperalgesia. If and how TRPV4 interacts with the microtubule and actin cytoskeleton at a molecular and functional level is not known.
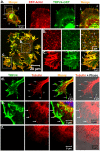
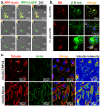
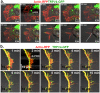
Methodology and principal findings: We investigated the interaction of TRPV4 with cytoskeletal components biochemically, cell biologically by observing morphological changes of DRG-neurons and DRG-neuron-derived F-11 cells, as well as functionally with calcium imaging. We find that TRPV4 physically interacts with tubulin, actin and neurofilament proteins as well as the nociceptive molecules PKCepsilon and CamKII. The C-terminus of TRPV4 is sufficient for the direct interaction with tubulin and actin, both with their soluble and their polymeric forms. Actin and tubulin compete for binding. The interaction with TRPV4 stabilizes microtubules even under depolymerizing conditions in vitro. Accordingly, in cellular systems TRPV4 colocalizes with actin and microtubules enriched structures at submembranous regions. Both expression and activation of TRPV4 induces striking morphological changes affecting lamellipodial, filopodial, growth cone, and neurite structures in non-neuronal cells, in DRG-neuron derived F11 cells, and also in IB4-positive DRG neurons. The functional interaction of TRPV4 and the cytoskeleton is mutual as Taxol, a microtubule stabilizer, reduces the Ca2+-influx via TRPV4.
Conclusions and significance: TRPV4 acts as a regulator for both, the microtubule and the actin. In turn, we describe that microtubule dynamics are an important regulator of TRPV4 activity. TRPV4 forms a supra-molecular complex containing cytoskeletal proteins and regulatory kinases. Thereby it can integrate signaling of various intracellular second messengers and signaling cascades, as well as cytoskeletal dynamics. This study points out the existence of cross-talks between non-selective cation channels and cytoskeleton at multiple levels. These cross talks may help us to understand the molecular basis of the Taxol-induced neuropathic pain development commonly observed in cancer patients.
Conflict of interest statement
Figures
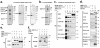


References
-
- Gao X, Wu L, O'Neil RG. Temperature-modulated diversity of TRPV4 channel gating: activation by physical stresses and phorbol ester derivatives through protein kinase C-dependent and -independent pathways. J Biol Chem. 2003;278:27129–37. - PubMed
-
- Mizuno A, Matsumoto N, Imai M, Suzuki M. Impaired osmotic sensation in mice lacking TRPV4. Am J Physiol Cell Physiol. 2003;285:C96–101. - PubMed
-
- Alessandri-Haber N, Yeh JJ, Boyd AE, Parada CA, Chen X, et al. Hypotonicity induces TRPV4-mediated nociception in rat. Neuron. 2003;39:497–511. - PubMed
-
- Strotmann R, Harteneck C, Nunnenmacher K, Schultz G, Plant TD. OTRPC4, a nonselective cation channel that confers sensitivity to extracellular osmolarity. Nat Cell Biol. 2000;2:695–702. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

