Identification of a novel snake peptide toxin displaying high affinity and antagonist behaviour for the α2-adrenoceptors
- PMID: 20659106
- PMCID: PMC3000660
- DOI: 10.1111/j.1476-5381.2010.00966.x
Identification of a novel snake peptide toxin displaying high affinity and antagonist behaviour for the α2-adrenoceptors
Abstract
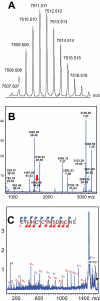
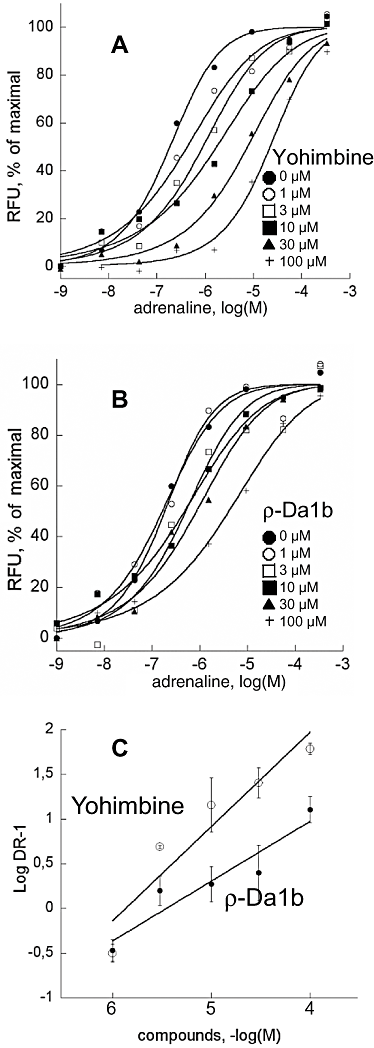
BACKGROUND AND PURPOSE Muscarinic and adrenergic G protein-coupled receptors (GPCRs) are the targets of rare peptide toxins isolated from snake or cone snail venoms. We used a screen to identify novel toxins from Dendroaspis angusticeps targeting aminergic GPCRs. These toxins may offer new candidates for the development of new tools and drugs. EXPERIMENTAL APPROACH In binding experiments with (3) H-rauwolscine, we studied the interactions of green mamba venom fractions with α(2) -adrenoceptors from rat brain synaptosomes. We isolated, sequenced and chemically synthesized a novel peptide, ρ-Da1b. This peptide was pharmacologically characterized using binding experiments and functional tests on human α(2)-adrenoceptors expressed in mammalian cells. KEY RESULTS ρ-Da1b, a 66-amino acid peptide stabilized by four disulphide bridges, belongs to the three-finger-fold peptide family. Its synthetic homologue inhibited 80% of (3) H-rauwolscine binding to the three α(2)-adrenoceptor subtypes, with an affinity between 14 and 73 nM and Hill slopes close to unity. Functional experiments on α(2A) -adrenoceptor demonstrated that ρ-Da1b is an antagonist, shifting adrenaline activation curves to the right. Schild regression revealed slopes of 0.97 and 0.67 and pA(2) values of 5.93 and 5.32 for yohimbine and ρ-Da1b, respectively. CONCLUSIONS AND IMPLICATIONS ρ-Da1b is the first toxin identified to specifically interact with α(2)-adrenoceptors, extending the list of class A GPCRs sensitive to toxins. Additionally, its affinity and atypical mode of interaction open up the possibility of its use as a new pharmacological tool, in the study of the physiological roles of α(2)-adrenoceptor subtypes.
British Journal of Pharmacology © 2010 The British Pharmacological Society. No claim to original French government works.
Figures

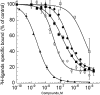
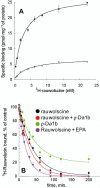
 ) or hα2C-adrenoceptor (3.1 µg, •) with ρ-Da1b. Inhibition of 3H-prazosin (1 nM) by ρ-Da1b in CHO membranes expressing hα1A-adrenoceptor (3.8 µg).
) or hα2C-adrenoceptor (3.1 µg, •) with ρ-Da1b. Inhibition of 3H-prazosin (1 nM) by ρ-Da1b in CHO membranes expressing hα1A-adrenoceptor (3.8 µg).
Similar articles
-
Isolation and pharmacological characterization of AdTx1, a natural peptide displaying specific insurmountable antagonism of the alpha1A-adrenoceptor.Br J Pharmacol. 2010 Jan 1;159(2):316-25. doi: 10.1111/j.1476-5381.2009.00532.x. Epub 2009 Dec 15. Br J Pharmacol. 2010. PMID: 20015090 Free PMC article.
-
G protein-coupled receptors, an unexploited animal toxin targets: Exploration of green mamba venom for novel drug candidates active against adrenoceptors.Toxicon. 2012 Mar 15;59(4):487-96. doi: 10.1016/j.toxicon.2011.03.009. Epub 2011 Mar 22. Toxicon. 2012. PMID: 21419153
-
Polypharmacology profiles and phylogenetic analysis of three-finger toxins from mamba venom: case of aminergic toxins.Biochimie. 2014 Aug;103:109-17. doi: 10.1016/j.biochi.2014.04.009. Epub 2014 May 1. Biochimie. 2014. PMID: 24793485
-
Muscarinic toxins from the green mamba.Pharmacol Ther. 2000 Feb;85(2):87-109. doi: 10.1016/s0163-7258(99)00064-9. Pharmacol Ther. 2000. PMID: 10722122 Review.
-
Selective targeting of G-protein-coupled receptor subtypes with venom peptides.Acta Physiol (Oxf). 2012 Feb;204(2):186-201. doi: 10.1111/j.1748-1716.2011.02305.x. Epub 2011 May 7. Acta Physiol (Oxf). 2012. PMID: 21481193 Review.
Cited by
-
Nature-Derived Peptides: A Growing Niche for GPCR Ligand Discovery.Trends Pharmacol Sci. 2019 May;40(5):309-326. doi: 10.1016/j.tips.2019.03.004. Epub 2019 Apr 5. Trends Pharmacol Sci. 2019. PMID: 30955896 Free PMC article. Review.
-
In-Depth Glyco-Peptidomics Approach Reveals Unexpected Diversity of Glycosylated Peptides and Atypical Post-Translational Modifications in Dendroaspis angusticeps Snake Venom.Int J Mol Sci. 2017 Nov 18;18(11):2453. doi: 10.3390/ijms18112453. Int J Mol Sci. 2017. PMID: 29156586 Free PMC article.
-
Ancestral protein resurrection and engineering opportunities of the mamba aminergic toxins.Sci Rep. 2017 Jun 2;7(1):2701. doi: 10.1038/s41598-017-02953-0. Sci Rep. 2017. PMID: 28578406 Free PMC article.
-
Adrenoceptor activity of muscarinic toxins identified from mamba venoms.Br J Pharmacol. 2011 Sep;164(2b):538-50. doi: 10.1111/j.1476-5381.2011.01468.x. Br J Pharmacol. 2011. PMID: 21557730 Free PMC article.
-
Cardiovascular Effects of Snake Toxins: Cardiotoxicity and Cardioprotection.Acta Naturae. 2021 Jul-Sep;13(3):4-14. doi: 10.32607/actanaturae.11375. Acta Naturae. 2021. PMID: 34707893 Free PMC article.
References
-
- Adem A, Asblom A, Johansson G, Mbugua PM, Karlsson E. Toxins from the venom of the green mamba Dendroaspis angusticeps that inhibit the binding of quinuclidinyl benzilate to muscarinic acetylcholine receptors. Biochim Biophys Acta. 1988;968:340–345. - PubMed
-
- Agrawal DK, Triggle CR, Daniel EE. Pharmacological characterization of the postsynaptic alpha adrenoceptors in vascular smooth muscle from canine and rat mesenteric vascular beds. J Pharmacol Exp Ther. 1984;229:831–838. - PubMed
-
- Antosova Z, Mackova M, Kral V, Macek T. Therapeutic application of peptides and proteins: parenteral forever? Trends Biotechnol. 2009;27:628–635. - PubMed
-
- Banerjee Y, Mizuguchi J, Iwanaga S, Kini RM. Hemextin AB complex, a unique anticoagulant protein complex from Hemachatus haemachatus (African Ringhals cobra) venom that inhibits clot initiation and factor VIIa activity. J Biol Chem. 2005;280:42601–42611. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

